Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
\(n_{CO_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
PTHH: C6H12O6 --men rượu--> 2C2H5OH + 2CO2
0,125<---------------------0,25<-------0,25
=> \(m_{C_2H_5OH}=0,25.46=11,5\left(g\right)\)
b) \(m_{C_6H_{12}O_6\left(pư\right)}=0,125.180=22,5\left(g\right)\)
=> \(m_{C_6H_{12}O_6\left(tt\right)}=\dfrac{22,5.100}{80}=28,125\left(g\right)\)
c) \(V_{C_2H_5OH}=\dfrac{11,5}{0,8}=14,375\left(ml\right)\)
=> \(V_{rượu}=\dfrac{14,375.100}{25}=57,5\left(ml\right)\)

\(n_{CO2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\)
Pt : \(C_6H_{12}O_6\xrightarrow[30-35^oC]{Menrượu}2C_2H_5OH+2CO_2\)
0,5 0,5
a) \(m_{C2H5OH}=0,5.46=23\left(g\right)\)
b) Pt : \(C_2H_5OH+O_2\xrightarrow[]{Mengiấm}CH_3COOH+H_2O\)
0,5 0,5
\(m_{CH3COOH\left(lt\right)}=0,5.60=30\left(g\right)\)
⇒ \(m_{CH3COOH\left(tt\right)}=30.80\%=24\left(g\right)\)
Chúc bạn học tốt
\(C_6H_{12}O_6\underrightarrow{t^o}2C_2H_5OH+2CO_2\uparrow\)(xt : men rượu )
0,5 0,5
\(n_{CO_2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\)
\(m_{C_2H_5OH}=0,5.46=23\left(g\right)\)
\(C_2H_5OH+O_2\underrightarrow{t^o}CH_3COOH+H_2O\) (men giấm )
0,5 0,5
\(m_{CH_3COOH}=0,5.60=30\left(g\right)\)
\(m_{CH_3COOHtt}=30.80\%=24\left(g\right)\)

a) n glucozo = 54/180 = 0,3(mol)
n glucozo pư = 0,3.80% = 0,24(mol)
$C_6H_{12}O_6 \xrightarrow{t^o} 2CO_2 +2 C_2H_5OH$
n C2H5OH = 2n glucozo = 0,48(mol)
m C2H5OH = 0,48.46 = 22,08(gam)
b)
$C_2H_5OH + O_2 \xrightarrow{men\ giấm} CH_3COOH + H_2O$
n CH3COOH = n C2H5OH = 0,48(mol)
C% CH3COOH = 0,48.60/500 .100% = 5,76%

\(n_{C_6H_{12}O_6}=\dfrac{1,8}{180}=0,01\left(mol\right)\)
PTHH: C6H12O6 --men rượu--> 2CO2 + 2C2H5OH
0,01---------------------->0,02----->0,02
=> \(\left\{{}\begin{matrix}V_{CO_2}=0,02.2.80\%.22,4=0,3584\left(l\right)\\m_{C_2H_5OH}=0,02.46.80\%=0,736\left(g\right)\end{matrix}\right.\)

a, \(n_{CH_3COOH}=0,2.1=0,2\left(mol\right)\)
PT: \(Mg+2CH_3COOH\rightarrow\left(CH_3COO\right)_2Mg+H_2\)
Theo PT: \(n_{Mg}=n_{H_2}=\dfrac{1}{2}n_{CH_3COOH}=0,1\left(mol\right)\)
\(\Rightarrow m=m_{Mg}=0,1.24=2,4\left(g\right)\)
\(V=V_{H_2}=0,1.22,4=2,24\left(l\right)\)
b, \(C_2H_5OH+O_2\underrightarrow{^{mengiam}}CH_3COOH+H_2O\)
Theo PT: \(n_{C_2H_5OH}=n_{CH_3COOH}=0,2\left(mol\right)\)
\(\Rightarrow m_{C_2H_5OH}=0,2.46=9,2\left(g\right)\)
\(\Rightarrow V_{ddC_2H_5OH}=\dfrac{9,2}{0,8}=11,5\left(ml\right)\)

\(V_{C_2H_5OH}=\dfrac{8.100000}{100}=8000\left(ml\right)\\ m_{C_2H_5OH}=8000.0,8=6400\left(ml\right)\\ n_{C_2H_5OH}=\dfrac{6400}{46}=\dfrac{3200}{23}\left(mol\right)\)
PTHH: C6H12O6 --to, men rượu--> 2C2H5OH + 2CO2
\(\dfrac{1600}{23}\)<----------------------------\(\dfrac{3200}{23}\)
\(n_{C_6H_{12}O_6}=\dfrac{\dfrac{3200}{23}}{95\%}=146,453\left(mol\right)\\ m_{C_6H_{12}O_6}=146,453.180=26361,54\left(g\right)\)
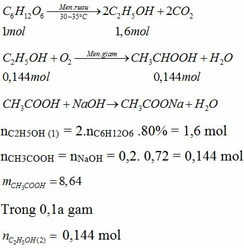

C6H12O6-lm->2C2H5OH+2CO2
0,25----------------0,5
n C2H5OH=0,5 mol
=>m =0,25.180.112%=50,4g
b) H=80%
m =50,4.\(\dfrac{100}{80}\)=63g