Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(nC_2H_5OH=\dfrac{2,9}{46}=0,06\left(mol\right)\)
\(C_2H_5OH+Na\rightarrow C_2H_5ONa+\dfrac{1}{2}H_2\)
0,06 0,06 0,06 0,03 (mol)
VH2 = 0,03.22,4= 0,672 (l)
V = m /D
=> V rượu etylic = 2,9 / 0,8 = 3,625 (ml)

đổi 120ml=0,12l
PTHH 2C2H5OH+ 2Na➝2C2H5ONa +H2
Theo đề nC2H5OH=\(\frac{0.12}{22.4}=0.005\left(mol\right)\)
Theo pt nH2=\(\frac{2}{1}.0.005=0.01\left(mol\right)\)
VH2=0.01.22.4=0.224(l)

a)
\(V_{C_2H_5OH}=\dfrac{96.20}{100}=19,2\left(ml\right)\)
=> \(m_{C_2H_5OH}=19,2.0,8=15,36\left(g\right)\)
b) \(n_{C_2H_5OH}=\dfrac{15,36}{46}=\dfrac{192}{575}\left(mol\right)\)
\(V_{H_2O}=20-19,2=0,8\left(ml\right)\)
=> \(m_{H_2O}=0,8.1=0,8\left(g\right)\)
=> \(n_{H_2O}=\dfrac{0,8}{18}=\dfrac{2}{45}\left(mol\right)\)
PTHH: 2C2H5OH + 2Na --> 2C2H5ONa + H2
\(\dfrac{192}{575}\)------------------------->\(\dfrac{96}{575}\)
2H2O + 2Na --> 2NaOH + H2
\(\dfrac{2}{45}\)----------------------->\(\dfrac{1}{45}\)
=> \(V_{H_2}=22,4.\left(\dfrac{96}{575}+\dfrac{1}{45}\right)=4,238\left(l\right)\)
C2H5OH + Na -- > C2H5OHNa + 1/2 H2
Na+H2O --- > NaOH + 1/2H2
Vr = 20x96/100 = 19,2ml = 0.0192 (l)
mC2H5OH = D.V = 19,2 x 0.8 = 15.36 (g)
nC2H5OH = m/M = 15.36 / 46 = 0.43 (mol)
=> nH2 = 0.215 (mol)
VH2O = 1 ml => mH2O = 1 (g)
=> nH2O = m/M = 1/18 = 0.056 (mol)
=> nH2 = 0.028 (mol)
nH2 = 0.215 + 0.028 = 0.243 (mol)
=> VH2 = 22.4 x 0,243 = 5,4432 (l)

\(a,V_{C_2H_5OH}=\dfrac{96.30}{100}=28,8\left(ml\right)\\ \rightarrow m_{C_2H_5OH}=28,8.0,8=23,04\left(ml\right)\\ \rightarrow n_{C_2H_5OH}=\dfrac{23,04}{46}=0,5\left(mol\right)\)
PTHH: 2C2H5OH + 2Na ---> 2C2H5ONa + H2
0,5----------------------------------->0,25
\(\rightarrow V_{H_2}=0,25.22,4=5,6\left(l\right)\)
b, \(n_{CH_3COOH}=\dfrac{36}{60}=0,6\left(mol\right)\)
PTHH: \(C_2H_5OH+CH_3COOH\xrightarrow[t^o]{H_2SO_{4\left(đ\right)}}CH_3COOC_2H_5+H_2O\)
LTL: 0,5 < 0,6 => CH3COOH dư
Theo pthh: nCH3COOH = nC2H5OH = 0,5 (mol)
=> meste = 0,5.88.70% = 30,8 (g)

a,VddC2H5OH=23+27=50(ml)
Đr=\(\dfrac{23}{50}\).100=46o
b,mC2H5OH=23.0,8=18,4(g)
nC2H5OH=\(\dfrac{18,4}{46}\)=0,4(mol)
PTHH: 2C2H5OH + 2K ---> 2C2H5OK + H2
0,4-------------------------------------->0,2
=> VH2 = 0,2.24 = 4,8 (l
a,VddC2H5OH=23+27=50(ml)
Đr=2350.100=46ob, mC2H5OH=23.0,8=18,4(g)
nC2H5OH=18,446=0,4(mol)a,
VddC2H5OH=23+27=50(ml)Đr=2350.100=46ob,
mC2H5OH=23.0,8=18,4(g)
nC2H5OH=18,446=0,4(mol)
PTHH: 2C2H5OH + 2K ---> 2C2H5OK + H2
0,4-------------->0,2
=> VH2 = 0,2.24 = 4,8 (l)

\(a,V_{ddC_2H_5OH}=23+27=50\left(ml\right)\\ Đ_r=\dfrac{23}{50}.100=46^o\\ b,m_{C_2H_5OH}=23.0,8=18,4\left(g\right)\\ n_{C_2H_5OH}=\dfrac{18,4}{46}=0,4\left(mol\right)\)
PTHH: 2C2H5OH + 2K ---> 2C2H5OK + H2
0,4-------------------------------------->0,2
=> VH2 = 0,2.24 = 4,8 (l)

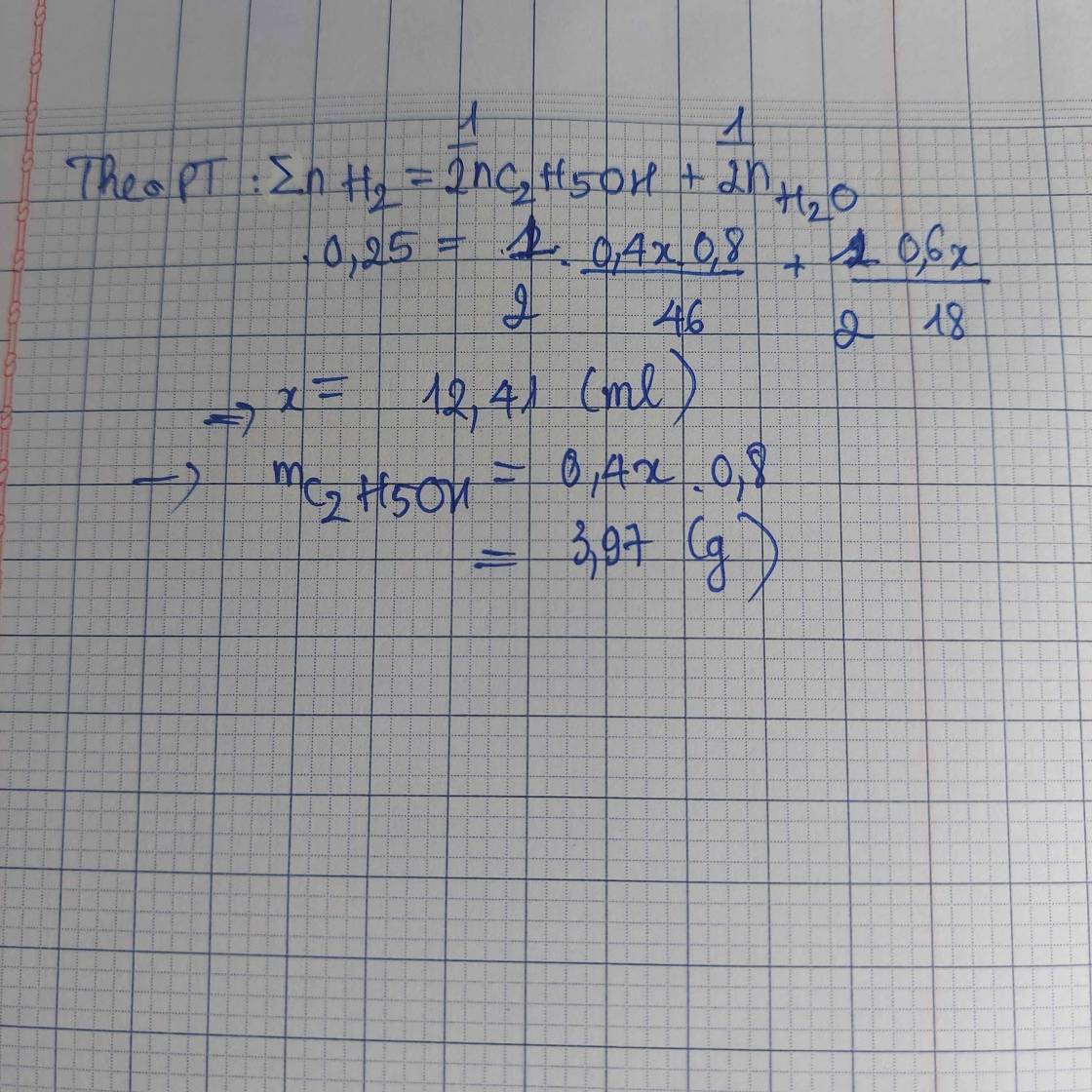
Vancol=200.45%=90ml=>m ancol=90.0,8=72g
=>n ancol=72/46=36/23 mol
VH2O=200-90=110ml=>mH2O=110g=>nH2O=55/9 mol
H2O + Na =>NaOH +1/2 H2
55/9 mol =>55/18 mol
C2H5OH + Na =>C2H5ONa +1/2 H2
36/23 mol =>18/23 mol
Tổng nH2=3,838 mol
=>VH2=85,974 lit
sai nhiều