Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.
Hòa tan hoàn toàn 1,1g hỗn hợp gồm Al, F thu được 0,896lit khí hidro ở đktc, tính khối lượng ban đầu

\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
\(n_{H_2}=\frac{0,896}{22,4}=0,04mol\)
\(\hept{\begin{cases}27a+56b=1,1\\1,5a+b=0,04\end{cases}}\)
\(\rightarrow\hept{\begin{cases}a=0,02\\b=0,01\end{cases}}\)
\(m_{Al}=0,02.27=0,54g\)
\(m_{Fe}=1,1-0,54=0,56g\)

Đáp án A
n H 2 = 0 , 2 ( m o l )
=> mhh= mFe + mAl
Bảo toàn electron:
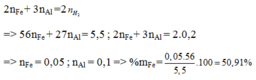

a) Gọi số mol Mg, Fe là a, b (mol)
=> 24a + 56b = 11,84
\(n_{HCl}=\dfrac{146.14\%}{36,5}=0,56\left(mol\right)\)
PTHH: Mg + 2HCl --> MgCl2 + H2
a--->2a--------->a----->a
Fe + 2HCl --> FeCl2 + H2
b-->2b-------->b------>b
=> 2a + 2b = 0,56
=> a = 0,12; b = 0,16
=> \(\left\{{}\begin{matrix}\%Mg=\dfrac{0,12.24}{11,84}.100\%=24,324\%\\\%Fe=\dfrac{0,16.56}{11,84}.100\%=75,676\%\end{matrix}\right.\)
b) \(n_{H_2}=a+b=0,28\left(mol\right)\)
=> \(V_{H_2}=0,28.22,4=6,272\left(l\right)\)
c) mdd sau pư = 11,84 + 146 - 0,28.2 = 157,28 (g)
=> \(\left\{{}\begin{matrix}C\%_{MgCl_2}=\dfrac{0,12.95}{157,28}.100\%=7,25\%\\C\%_{FeCl_2}=\dfrac{0,16.127}{157,28}.100\%=12,92\%\end{matrix}\right.\)

a) \(n_{AlCl_3}=\dfrac{6,675}{133,5}=0,05\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
0,05<-----------0,05---->0,075
=> \(\%Al=\dfrac{0,05.27}{14,15}.100\%=9,54\%\)
=> \(\%Cu=\dfrac{14,15-0,05.27}{14,15}.100\%=90,46\%\)
b) \(V_{H_2}=0,075.22,4=1,68\left(l\right)\)
c) \(n_{Cu}=\dfrac{14,15-0,05.27}{64}=0,2\left(mol\right)\)
PTHH: 4Al + 3O2 --to--> 2Al2O3
0,05->0,0375
2Cu + O2 --to--> 2CuO
0,2-->0,1
=> \(V_{O_2}=\left(0,1+0,0375\right).22,4=3,08\left(l\right)\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\\ m_{AlCl_3}=6,675\left(mol\right)\\ n_{AlCl_3}=\dfrac{6,675}{133,5}=0,05\left(mol\right)\\ \Rightarrow n_{Al}=n_{AlCl_3}=0,05\left(mol\right)\\ \Rightarrow m_A=0,05.27=1,35\left(g\right);m_{Cu}=14,15-1,35=12,8\left(g\right)\\ \%m_{Cu}=\dfrac{12,8}{14,15}.100\approx90,459\%\\ \Rightarrow\%m_{Al}\approx9,541\%\\ b,n_{Cu}=\dfrac{12,8}{64}=0,2\left(mol\right)\\ n_{H_2}=\dfrac{3}{2}.n_{Al}=\dfrac{3}{2}.0,05=0,075\left(mol\right)\\ \Rightarrow V=V_{H_2\left(đktc\right)}=0,075.22,4=1,68\left(l\right)\\ 4Al+3O_2\rightarrow\left(t^o\right)2Al_2O_3\\ 2Cu+O_2\rightarrow\left(t^o\right)2CuO\\ n_{O_2}=\dfrac{3}{4}.n_{Al}+\dfrac{1}{2}.n_{Cu}=\dfrac{3}{4}.0,05+\dfrac{1}{2}.0,2=0,0875\left(mol\right)\)
\(\Rightarrow V_{O_2\left(đktc\right)}=0,0875.22,4=1,96\left(l\right)\)

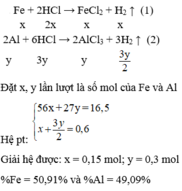

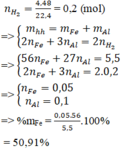

\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
\(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
\(n_{H_2}=\frac{0,896}{22,4}=0,04mol\)
\(\hept{\begin{cases}27a+56b=1,1\\1,5a+b=0,04\end{cases}}\)
\(\Rightarrow\hept{\begin{cases}a=0,02\\b=0,01\end{cases}}\)
\(m_{Al}=0,02.27=0,54g\)
\(m_{Fe}=1,1-0,54=0,56g\)