Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
a) 2Al + 6HCl --> 2AlCl3 + 3H2
b) \(n_{H_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
2Al + 6HCl --> 2AlCl3 + 3H2
\(\dfrac{0,4}{3}\)<--------------------0,2
=> Al dư
\(m_{Al\left(dư\right)}=\left(0,2-\dfrac{0,4}{3}\right).27=1,8\left(g\right)\)

2Al+6HCl->2AlCl3+3H2
0,05----0,15------------------0,075 mol
n H2=\(\dfrac{1,68}{22,4}\)=0,075 mol
=>m Al=0,05.27=1,35g
=>HCl dư =>m HCl=0,1.36,5=3,65g

\(n_{Al}=\dfrac{m_{Al}}{M_{Al}}=\dfrac{5,4}{27}=0,2mol\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
0,2 0,2 0,3 ( mol )
\(V_{H_2}=n_{H_2}.22,4=0,3.22,4=6,72l\)
\(m_{AlCl_3}=n_{AlCl_3}.M_{AlCl_3}=0,2.133,5=26,7g\)

a)
\(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
0,2----------->0,2----->0,3
=> \(V_{H_2}=0,3.22,4=6,72\left(l\right)\)
b) \(m_{AlCl_3}=0,2.133,5=26,7\left(g\right)\)
c)
PTHH: Zn + H2SO4 --> ZnSO4 + H2
0,3<----------------0,3
=> \(m_{H_2SO_4}=0,3.98=29,4\left(g\right)\)
\(a,n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
PTHH:
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
0,2--------------->0,2------->0,3
\(V_{H_2}=0,3.22,4=6,72\left(l\right)\\ b,m_{AlCl_3}=0,2.133,5=26,7\left(g\right)\)
c, PTHH:
\(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\)
0,2<------------------0,2
\(m_{H_2SO_4}=0,2.98=19,6\left(g\right)\)

a, \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
b, \(n_{Al}=\dfrac{8,1}{27}=0,3\left(mol\right)\)
\(n_{H_2}=\dfrac{3}{2}n_{Al}=0,45\left(mol\right)\Rightarrow V_{H_2}=0,45.22,4=10,08\left(l\right)\)
c, \(n_{AlCl_3}=n_{Al}=0,3\left(mol\right)\Rightarrow m_{AlCl_3}=0,3.133,5=40,05\left(g\right)\)
d, \(n_{Fe_2O_3}=\dfrac{32}{160}=0,2\left(mol\right)\)
PT: \(Fe_2O_3+3H_2\underrightarrow{t^o}2Fe+3H_2O\)
Có: \(\dfrac{0,2}{1}>\dfrac{0,45}{3}\) → Fe2O3 dư.
\(n_{Fe}=\dfrac{2}{3}n_{H_2}=0,3\left(mol\right)\Rightarrow m_{Fe}=0,3.56=16,8\left(g\right)\)

a) PTHH: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(2mol\) \(6mol\) \(2mol\) \(3mol\)
\(0,27\) \(x\) \(y\) \(z\)
b) ta có: \(n_{Al}=\dfrac{m_{Al}}{M_{Al}}=\dfrac{7,3}{27}=0,27\left(mol\right)\)
theo PT: \(n_{Al}=n_{AlCl_3}=0,27\left(mol\right)\)
\(\Rightarrow m_{AlCl_3}=n_{AlCl_3}.M_{AlCl_3}=0,27.133,5=36,045\left(g\right)\)
c) ta có: \(n_{H_2}=\dfrac{m_{H_2}}{M_{H_2}}=\) \(\dfrac{0,27.3}{2}=0,405\left(mol\right)\)
\(\Rightarrow V_{H_2\left(đktc\right)}=n_{H_2}.22,4=0,405.22,4=9,072\left(l\right)\)

\(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right);n_{HCl}=\dfrac{3,65}{36,5}=0,1\left(mol\right)\\ a,Mg+2HCl\rightarrow MgCl_2+H_2\\ Vì:\dfrac{0,1}{2}< \dfrac{0,1}{1}\\ \Rightarrow Mgdư\\ \Rightarrow n_{Mg\left(p.ứ\right)}=n_{MgCl_2}=n_{H_2}=\dfrac{0,1}{2}=0,05\left(mol\right)\\ n_{Mg\left(dư\right)}=0,1-0,05=0,05\left(mol\right)\\ \Rightarrow m_{Mg\left(dư\right)}=0,05.24=1,2\left(g\right)\\ b,m_{MgCl_2}=95.0,05=4,75\left(g\right)\\ c,V_{H_2\left(đktc\right)}=0,05.22,4=1,12\left(l\right)\)

a. 2Al + 6HCl -> 2AlCl3 + 3H2
b. nAl = \(\dfrac{8.1}{27}=0,3\left(mol\right)\)=> \(n_{H_2}=\dfrac{3}{2}.0,3=0,45\left(mol\right)\)
\(V_{H_2}=0,45.22,4=10,08\left(mol\right)\)
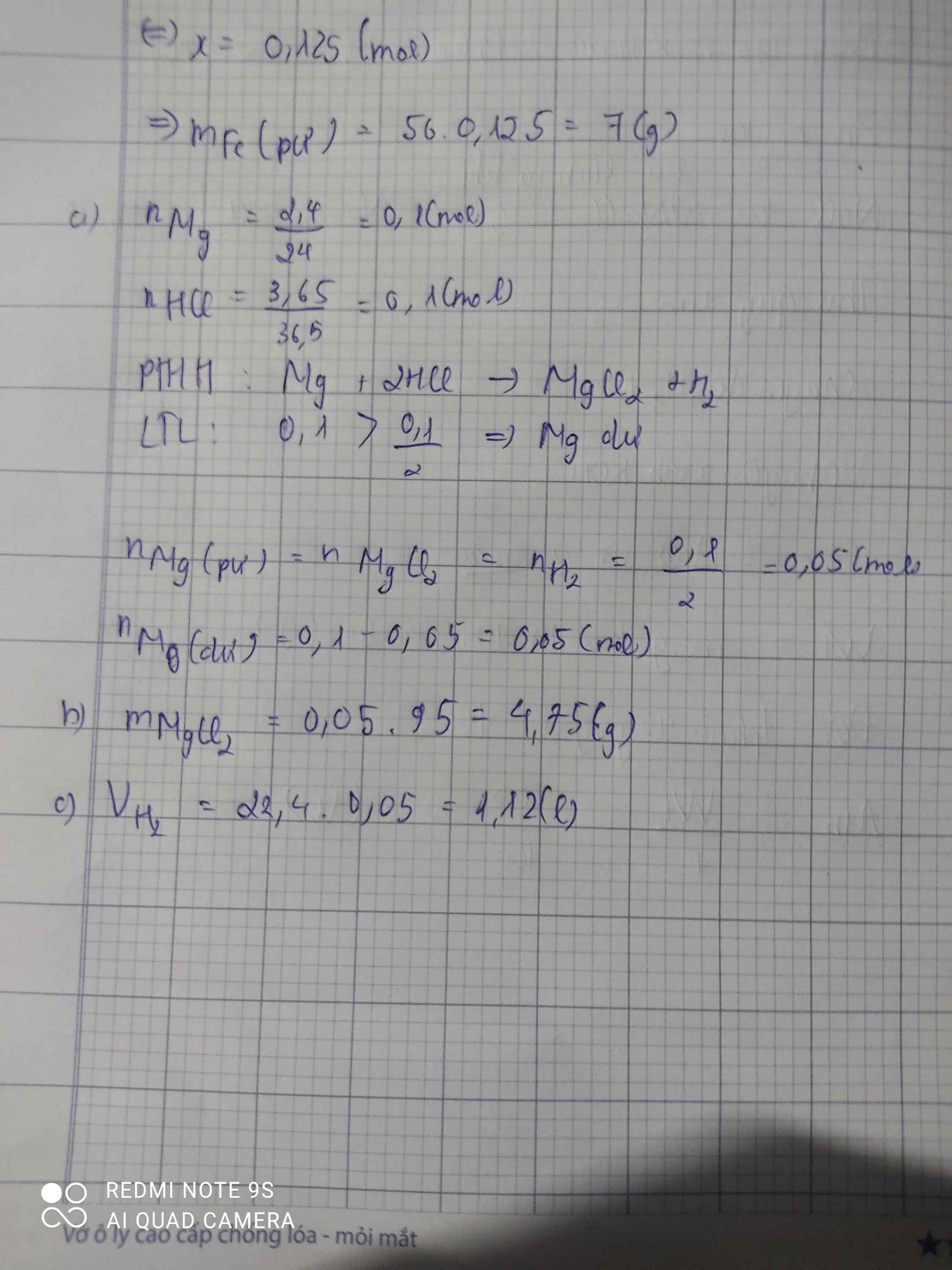
a: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(n_{Al}=\dfrac{5.4}{27}=0.2\left(mol\right)\)
\(n_{HCl}=\dfrac{10.95}{36.5}=0.3\left(mol\right)\)
Vì \(\dfrac{0.2}{2}>\dfrac{0.3}{6}\) nên Al dư và dư 0,05 mol
b: \(n_{AlCl_3}=0.9\left(mol\right)\)
\(m_{AlCl_3}=0.9\cdot136.5=122.85\left(g\right)\)