
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Bài 4:
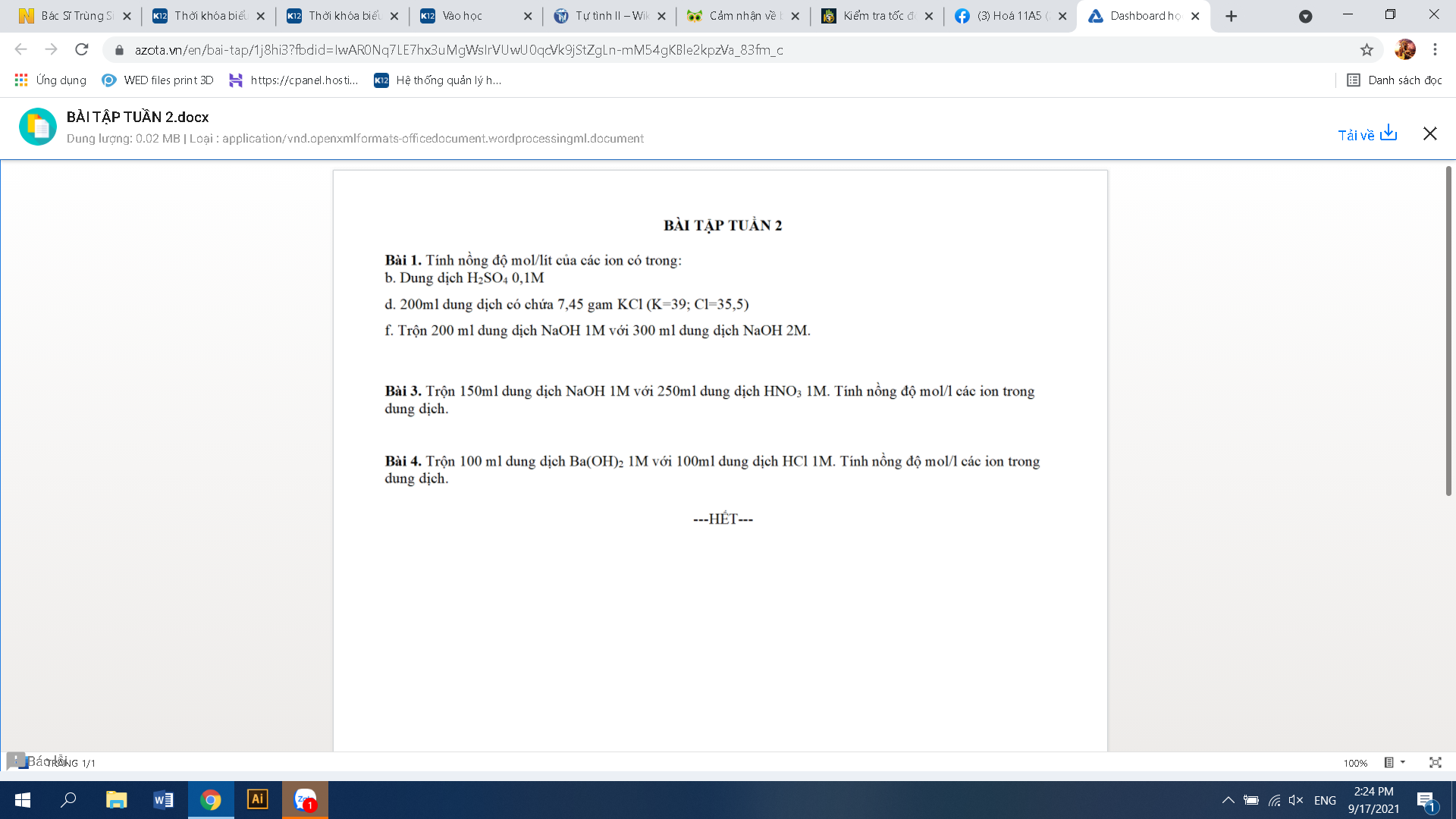
\(n_{Ba\left(OH\right)_2}=1.0,1=0,1\left(mol\right)\\ n_{HCl}=1.0,1=0,1\left(mol\right)\\ Ba\left(OH\right)_2+2HCl\rightarrow BaCl_2+2H_2O\\ Vì:\dfrac{0,1}{2}< \dfrac{0,1}{1}\Rightarrow Ba\left(OH\right)_2dư\\ n_{BaCl_2}=n_{Ba\left(OH\right)_2\left(p.ứ\right)}=\dfrac{0,1}{2}=0,05\left(mol\right)\\ n_{Ba\left(OH\right)_2\left(dư\right)}=0,1-0,05=0,05\left(mol\right)\\ \left[OH^-\left(dư\right)\right]=\left[Ba\left(OH\right)_2\left(dư\right)\right]=\dfrac{0,05}{0,1+0,1}=0,25\left(M\right)\\ \left[Cl^-\right]=2.\left[BaCl_2\right]=2.\dfrac{0,05}{0,1+0,2}=0,5\left(M\right)\\ \left[Ba^{2+}\right]=\dfrac{0,25+0,5}{2}=0,375\left(M\right)\)




Câu 3:
\(n_{CO_2}=\dfrac{0,44}{44}=0,01\left(mol\right)\)
\(n_{H_2O}=\dfrac{0,18}{18}=0,01\left(mol\right)\)
Bảo toàn C: nC(A) = 0,01 (mol)
Bảo toàn H: nC(A) = 2.0,01 = 0,02 (mol)
=> \(n_O=\dfrac{0,3-0,01.12-0,02.1}{16}=0,01\left(mol\right)\)
nC : nH : nO = 0,01 : 0,02 : 0,01 = 1:2:1
=> CTHH: (CH2O)n
Có\(n_{O_2}=\dfrac{0,32}{32}=0,01\left(mol\right)=>M_A=\dfrac{0,3}{0,01}=30\left(g/mol\right)\)
=> n = 1
=> CTHH: CH2O
Câu 4:
\(n_{NO_2}=\dfrac{5,152}{22,4}=0,23\left(mol\right)\)
PTHH: Cu + 4HNO3 --> Cu(NO3)2 + 2NO2 + 2H2O
_____a---------------------------------->2a
Fe + 6HNO3 --> Fe(NO3)3 + 3NO2 + 3H2O
b---------------------------------->3b
=> \(\left\{{}\begin{matrix}64a+56b=5,36\\2a+3b=0,23\end{matrix}\right.=>\left\{{}\begin{matrix}a=0,04\\b=0,05\end{matrix}\right.\)
=> \(\left\{{}\begin{matrix}\%Cu=\dfrac{0,04.64}{5,36}.100\%=47,76\%\\\%Fe=\dfrac{0,05.56}{5,36}.100\%=52,24\%\end{matrix}\right.\)



$C + O_2 \xrightarrow{t^o} CO_2$
$2C + O_2 \xrightarrow{t^o} 2CO$
Gọi $n_{CO_2} = a(mol) ; n_{CO} =b (mol)$
Ta có: $M_B = \dfrac{44a + 28b}{a + b} = 15.2 = 30(1)$
Theo PTHH : $n_{O_2} = a + 0,5b = \dfrac{2,88}{32} = 0,09(2)$
Từ (1)(2) suy ra : a = 0,02 ; b = 0,14
$\%V_{CO_2} = \dfrac{0,02}{0,02 + 0,14}.100\% = 12,5\%$
$\%V_{CO} = 100\% - 12,5\% = 87,5\%$

a)
P1:
\(n_{Br_2}=\dfrac{80.20\%}{160}=0,1\left(mol\right)\)
PTHH: C2H4 + Br2 --> C2H4Br2
0,1<--0,1
=> \(n_{C_2H_4\left(P_1\right)}=0,1\left(mol\right)\)
=> \(m_{C_3H_8\left(P_1\right)}=\dfrac{12,2}{2}-0,1.28=3,3\left(g\right)\)
=> \(n_{C_3H_8\left(P_1\right)}=\dfrac{3,3}{44}=0,075\left(mol\right)\)
=> \(V=\left(0,1.2+0,075,2\right).22,4=7,84\left(l\right)\)
\(\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,1}{0,1+0,075}.100\%=57,143\%\\\%V_{C_3H_8}=\dfrac{0,075}{0,1+0,075}.100\%=42,857\%\end{matrix}\right.\)
b) P2 \(\left\{{}\begin{matrix}C_2H_4:0,1\left(mol\right)\\C_3H_8:0,075\left(mol\right)\end{matrix}\right.\)
Bảo toàn C: \(n_{CO_2}=0,425\left(mol\right)\) => \(n_{BaCO_3}=0,425\left(mol\right)\)
Bảo toàn H: \(n_{H_2O}=0,5\left(mol\right)\)
Xét \(\Delta m=m_{CO_2}+m_{H_2O}-m_{BaCO_3}=0,425.44+0,5.18-0,425.197=-56,025\left(g\right)\)
=> khối lượng dd sau pư giảm 56,025 gam





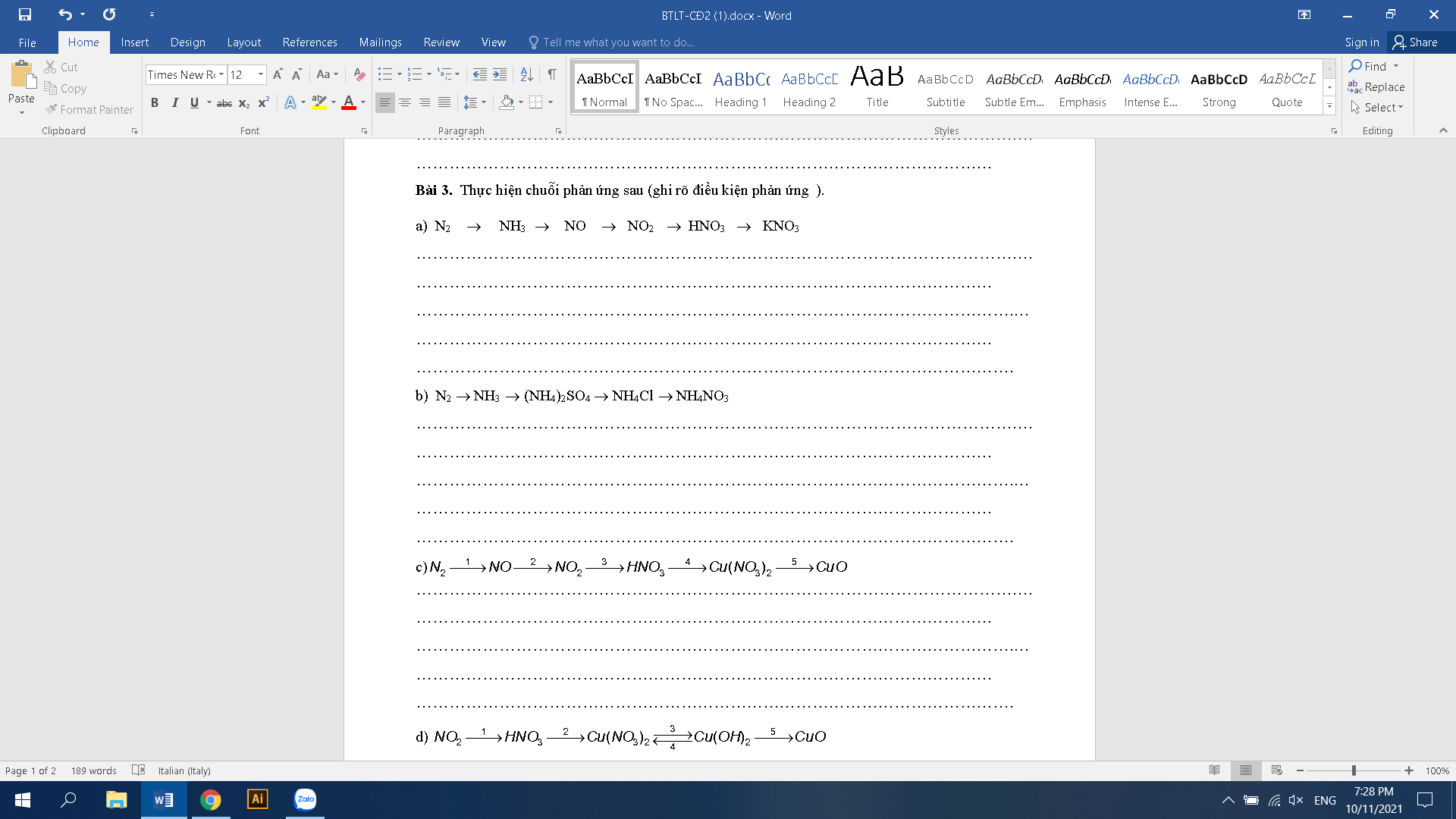



3.