
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


$a) MnO_2 + 4HCl \xrightarrow{t^o} MnCl_2 + Cl_2 + 2H_2O$
$Cl_2 + Cu \xrightarrow{t^o} CuCl_2$
$CuCl_2 + 2NaOH \to Cu(OH)_2 + 2NaCl$
$2NaCl + 2H_2O \xrightarrow{đpdd, cmn} 2NaOH + H_2 + Cl_2$
$2NaOH + Cl_2 \to NaCl + NaClO + H_2O$
$NaClO + HCl \to NaCl + HClO$
b)
$2KMnO_4 + 16HCl \to 2KCl + 2MnCl_2 + 5Cl_2 + 8H_2O$
$Cl_2 + H_2 \xrightarrow{ánh\ sáng} 2HCl$
$HCl + KOH \to KCl + H_2O$
$2KCl + 2H_2O \xrightarrow{đpdd, cmn} 2KOH + H_2 + Cl_2$
$3Cl_2 + 2Fe \xrightarrow{t^o} 2FeCl_3$
$FeCl_3 + 3AgNO_3 \to Fe(NO_3)_3 + 3AgCl$

\(n_{Fe}=\dfrac{5.6}{56}=0.1\left(mol\right)\)
\(Fe+\dfrac{3}{2}Cl_2\underrightarrow{^{^{t^0}}}FeCl_3\)
\(0.1.......0.15..........0.1\)
\(V_{Cl_2}=0.15\cdot22.4=3.36\left(l\right)\)
\(m_{FeCl_3}=0.1\cdot162.5=16.25\left(g\right)\)

1. \(2Ca+O_2\underrightarrow{t^o}2CaO\)
2. \(2Na+Cl_2\underrightarrow{t^o}2NaCl\)
3. \(2Fe+3Cl_2\underrightarrow{t^o}2FeCl_3\)
4. \(Na_2O+H_2O\rightarrow2NaOH\)
5. \(2Al+3S\underrightarrow{t^o}Al_2S_3\)
6. \(2SO_2+O_2\underrightarrow{t^o}2SO_3\)
7. \(Fe+2FeCl_3\rightarrow3FeCl_2\)
8. \(FeCl_2+2NaOH\rightarrow Fe\left(OH\right)_{2\downarrow}+2NaCl\)
9. \(Mg+2HCl\rightarrow MgCl_2+H_2\)
10. \(2Fe\left(OH\right)_3\underrightarrow{t^o}Fe_2O_3+3H_2O\)
11. \(P_2O_5+3H_2O\rightarrow2H_3PO_4\)
12. \(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
13. Bạn xem lại đề câu này nhé!
14. \(H_2+\dfrac{1}{2}O_2\underrightarrow{t^o}H_2O\)
15. \(K+H_2O\rightarrow KOH+\dfrac{1}{2}H_2\)
16. \(2FeCl_2+Cl_2\underrightarrow{t^o}2FeCl_3\)
Bạn tham khảo nhé!
1, 2Ca + O2---> 2CaO
2, 2Na + Cl2---> 2NaCl
3, 2Fe + 3Cl2---> 2FeCl3
4, Na2O + H2O---> 2NaOH
5, 2Al + 3S---> Al2S3
6, 2SO2 + O2---> 2SO3
7, Fe + 2FeCl3---> 3FeCl2
8, FeCl2 + 2NaOH---> Fe(OH)2 + 2NaCl
9, Mg + 2HCl---> MgCl2 + H2
10, 2Fe(OH)3 ---> Fe2O3 + 2H2O
11, P2O5 + 3H2O---> 2H3PO4
12, 4P + 5O2---> 2P2O5
13, NaHCO3 + N2CO3---> H2O + O2 ( cái này mk nhớ là ko có, bạn kiểm tra lại xe có sai ko nhé )
14, 2H2 + O2---> 2H2O
15, 2K + 2H2O---> 2KOH + H2
16, 2FeCl2 + Cl2---> 2FeCl3
Chúc bạn học tốt!!! ![]()

1. CaO + 2HCl ---> CaCl2 + H2O (1 : 2 : 1 : 1)
2. 2Fe + 3Cl2 ---to---> 2FeCl3 (2 : 3 : 2)
3. Zn + 2HCl ---> ZnCl2 + H2 (1 : 2 : 1 : 1)
4. 3Ba(OH)2 + 2FeCl3 ---> 2Fe(OH)3 + 3BaCl2 (3 : 2 : 2 : 3)
5. 4Al + 3O2 ---to---> 2Al2O3 (4 : 3 : 2)

1) \(2Fe+3Cl_2\underrightarrow{t^o}2FeCl_3\)
2) \(Ba\left(NO_3\right)_2+Na_2CO_3\rightarrow BaCO_3\downarrow+2NaNO_3\)
3) \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
4) \(3H_2SO_4+2Fe\left(OH\right)_3\rightarrow Fe_2\left(SO_4\right)_3+6H_2O\)
5) \(Fe_2O_3+3CO\underrightarrow{t^o}2Fe+3CO_2\)
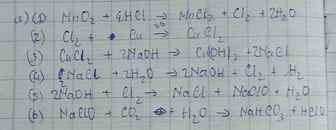
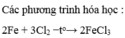
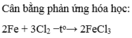

2Fe + 3Cl2 -> 2FeCl3
2Fe + 3Cl2 -> 2FeCl3