Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

PT: \(\left(C_{17}H_{35}COO\right)_3C_3H_5+3NaOH\underrightarrow{t^o}3C_{17}H_{35}COONa+C_3H_5\left(OH\right)_3\)
Ta có: \(n_{\left(C_{17}H_{35}COO\right)_3C_3H_5}=\dfrac{4500}{890}=\dfrac{450}{89}\left(mol\right)\)
Theo PT: \(n_{C_{17}H_{35}COONa}=3n_{\left(C_{17}H_{35}COO\right)_3C_3H_5}=\dfrac{1350}{89}\left(mol\right)\)
\(\Rightarrow m_{C_{17}H_{35}COONa}=\dfrac{1350}{89}.306\approx4641,6\left(g\right)\)
\(\Rightarrow m_{xp}=\dfrac{4641,6}{60\%}=7736\left(g\right)\)
Bạn tham khảo nhé!

PTHH: \(\left(RCOO\right)_3C_3H_5+3NaOH\rightarrow3RCOONa+C_3H_5\left(OH\right)_3\)
Áp dụng ĐLBTKL:
\(m_{hhaxit.béo}=8,58+1,2-0,368=9,412\left(kg\right)\\ \rightarrow m_{xà.phòng}=9,412.\left(100\%-60\%\right)=3,7648\left(kg\right)\)

\(n_{NaOH}=\dfrac{12}{40}=0,3\left(mol\right)\\ PTHH:\left(RCOO\right)_3C_3H_5+3NaOH\rightarrow3RCOONa+C_3H_5\left(OH\right)_3\\ \Rightarrow n_{C_3H_5\left(OH\right)_3}=\dfrac{1}{3}n_{NaOH}=0,1\left(mol\right)\\ \Rightarrow m_{C_3H_5\left(OH\right)_3}=0,1\cdot92=9,2\left(g\right)\\ \Rightarrow m_{RCOONa}=m_{\left(RCOO\right)_3C_3H_5}+m_{NaOH}-m_{C_3H_5\left(OH\right)_3}=89+12-9,2=91,8\left(g\right)\)

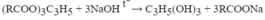
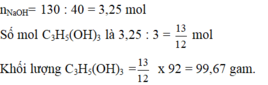

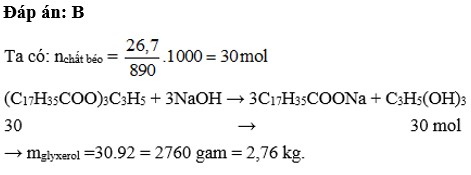
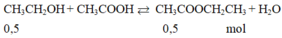
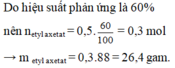
\(\left(C_{17}H_{35}COO\right)_3C_3H_5+3NaOH\rightarrow3C_{17}H_{35}COONa+C_3H_5\left(OH\right)_3\\ n_{\left(C_{17}H_{35}COO\right)_3C_3H_5}=\dfrac{8,9}{890}=0,01\left(mol\right)\\ n_{C_{17}H_{35}COONa}=3n_{\left(C_{17}H_{35}COO\right)_3C_3H_5}=0,01.3=0,03\left(mol\right)\\ \Rightarrow m_{C_{17}H_{35}COONa}=0,03.306=9,18\left(kg\right)\\ \Rightarrow m_{xp}=\dfrac{9,18}{60\%}=15,3\left(kg\right)\)