Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Gọi số mol H2, O2 là a, b (mol)
=> \(\left\{{}\begin{matrix}a+b=\dfrac{22,4}{22,4}=1\\M_B=\dfrac{2a+32b}{a+b}=5,5.2=11\left(g/mol\right)\end{matrix}\right.\)
=> a = 0,7 (mol); b = 0,3 (mol)
PTHH: 2H2 + O2 --to--> 2H2O
Xét tỉ lệ: \(\dfrac{0,7}{2}>\dfrac{0,3}{1}\) => H2 dư, O2 hết
PTHH: 2H2 + O2 --to--> 2H2O
0,6<--0,3------->0,6
=> \(\left\{{}\begin{matrix}m_{H_2O}=0,6.18=10,8\left(g\right)\\m_{H_2\left(dư\right)}=\left(0,7-0,6\right).2=0,2\left(g\right)\end{matrix}\right.\)

a)
C2H4 + 3O2 --to--> 2CO2 + 2H2O
2C3H6 + 9O2 --to--> 6CO2 + 6H2O
C2H4O2 + 2O2 --to--> 2CO2 + 2H2O
b)
Quy đổi C2H4, C3H6 thành CnH2n
hh chứa \(\left\{{}\begin{matrix}C_nH_{2n}:a\left(mol\right)\\C_2H_4O_2:b\left(mol\right)\end{matrix}\right.\)
=> 14an + 60b = 13 (1)
PTHH: CnH2n + \(\dfrac{3n}{2}\)O2 --to--> nCO2 + nH2O
a--->1,5an-------->an
C2H4O2 + 2O2 --to--> 2CO2 + 2H2O
b----->2b---------->2b
=> \(n_{CO_2}=an+2b=\dfrac{15,68}{22,4}=0,7\left(mol\right)\) (2)
(1)(2) => an = 0,5; b = 0,1 (mol)
\(V=22,4.\left(1,5an+2b\right)=21,28\left(l\right)\)
c) \(\%m_{C_2H_4O_2}=\dfrac{60.0,1}{13}.100\%=46,154\%\)

\(n_{O_2}=\dfrac{89.6}{22.4}=4\left(mol\right)\)
\(n_{H_2O}=3a\left(mol\right)\)
\(n_{CO_2}=a\left(mol\right)\)
\(2H_2+O_2\underrightarrow{^{^{t^0}}}2H_2O\)
\(2CO+O_2\underrightarrow{^{^{t^0}}}2CO_2\)
\(n_{O_2}=1.5a+0.5a=4\left(mol\right)\)
\(\Leftrightarrow a=2\)
\(n_{H_2}=3\left(mol\right),n_{CO}=1\left(mol\right)\)
\(\%V_{H_2}=\dfrac{3}{4}\cdot100\%=75\%\)
\(\%V_{CO}=25\%\)
\(\%m_{H_2}=\dfrac{3\cdot2}{3\cdot2+1\cdot28}\cdot100\%=17.64\%\)
\(\%m_{CO}=100-17.64=82.36\%\)

a, \(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
\(C_2H_4+3O_2\underrightarrow{t^o}2CO_2+2H_2O\)
\(2C_2H_2+5O_2\underrightarrow{t^o}4CO_2+2H_2O\)
b, Sửa đề: 17,9 (l) → 17,92 (l)
Ta có: \(n_{CO_2}=\dfrac{17,92}{22,4}=0,8\left(mol\right)=n_C\)
\(n_{H_2O}=\dfrac{18}{18}=1\left(mol\right)\Rightarrow n_H=1.2=2\left(mol\right)\)
⇒ mA = mC + mH = 0,8.12 + 2.1 = 11,6 (g)
Theo ĐLBT KL, có: mA + mO2 = mCO2 + mH2O
⇒ mO2 = 0,8.44 + 18 - 11,6 = 41,6 (g)
\(\Rightarrow n_{O_2}=\dfrac{41,6}{32}=1,3\left(mol\right)\Rightarrow V_{O_2}=1,3.22,4=29,12\left(l\right)\)

Giả sử các khí được đo ở điều kiện sao cho 1 mol khí chiếm thể tích 1 lít
Gọi số mol CH4, C2H6 là a, b (mol)
=> \(a+b=\dfrac{25}{1}=25\left(mol\right)\) (1)
\(n_{O_2}=\dfrac{95}{1}=95\left(mol\right)\)
PTHH: CH4 + 2O2 --to--> CO2 + 2H2O
a---->2a---------->a
2C2H6 + 7O2 --to--> 4CO2 + 6H2O
b------>3,5b-------->2b
=> \(\left\{{}\begin{matrix}n_{O_2\left(dư\right)}=95-2a-3,5b\left(mol\right)\\n_{CO_2}=a+2b\left(mol\right)\end{matrix}\right.\)
=> \(95-a-1,5b=\dfrac{60}{1}=60\)
=> a + 1,5b = 35 (2)
(1)(2) => a = 5; b = 20
=> \(\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{5}{25}.100\%=20\%\\\%V_{C_2H_6}=\dfrac{20}{25}.100\%=80\%\end{matrix}\right.\)
\(\overline{M}_A=\dfrac{5.16+20.30}{5+20}=27,2\left(g/mol\right)\)
\(\overline{M}_B=20,5.2=41\left(g/mol\right)\)
=> \(d_{A/B}=\dfrac{27,2}{41}\approx0,663\)

\(a)\\ 2CO + O_2 \xrightarrow{t^o} 2CO\\ 2H_2 + O_2 \xrightarrow{t^o} 2H_2O\\ n_{H_2} = n_{H_2O} = \dfrac{1,8}{18} = 0,1(mol)\\ \)
Theo PTHH :
\(2n_{O_2} = n_{CO} + n_{H_2}\\ \Leftrightarrow 2.\dfrac{3,36}{22,4} = n_{CO} + 0,1\\ \Leftrightarrow n_{CO} = 0,2(mol)\\ \%V_{H_2} = \dfrac{0,1}{0,1+ 0,2}.100\% = 33,33\%\\ \%V_{CO} = 100\%-33,33\% = 66,67\%\\ c) Cách\ 1 :\\ n_{CO_2} = n_{CO} = 0,2(mol)\\ m_{CO_2} = 0,2.44 = 8,8(gam)\\ Cách\ 2 : \\ m_{hh} = m_{CO} + m_{H_2} = 0,2.28 + 0,1.2 = 5,8(gam) \)
Bảo toàn khối lượng :
\(m_{hh} + m_{O_2} = m_{H_2O} + m_{CO_2}\\ \Rightarrow m_{CO_2} = 5,8 + 0,15.32 - 1,8 = 8,8(gam)\)

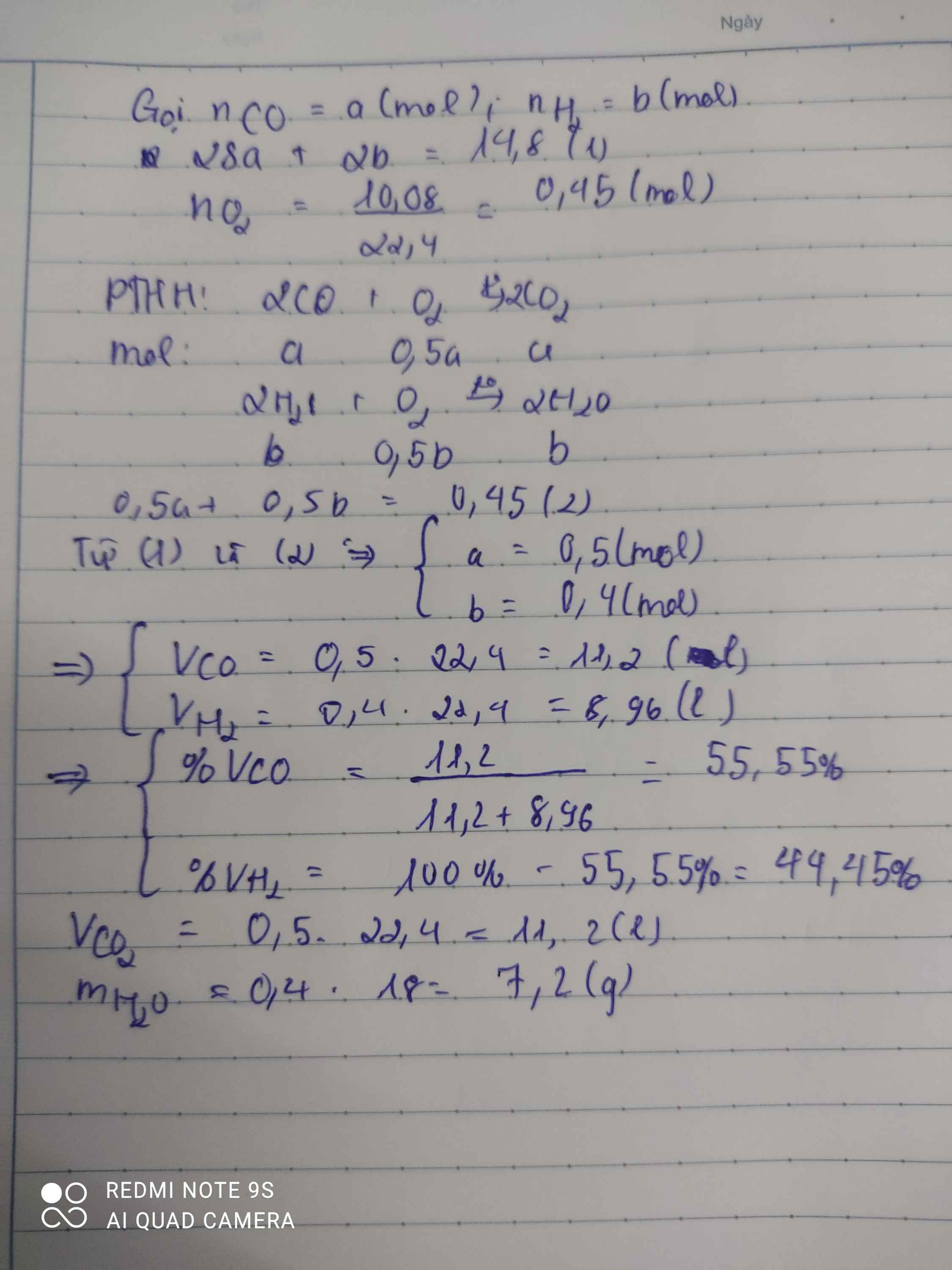
a)
2CO + O2 --to--> 2CO2
2H2 + O2 --to--> 2H2O
b) \(n_{H_2O}=\dfrac{12,6}{18}=0,7\left(mol\right)\); \(n_{CO_2}=\dfrac{13,44}{22,4}=0,6\left(mol\right)\)
PTHH: 2CO + O2 --to--> 2CO2
0,6<--0,3<------0,6
2H2 + O2 --to--> 2H2O
0,7<--0,35<------0,7
=> \(\left\{{}\begin{matrix}V_{CO}=0,6.22,4=13,44\left(l\right)\\V_{H_2}=0,7.22,4=15,68\left(l\right)\end{matrix}\right.\)
VO2 = (0,3 + 0,35).22,4 = 14,56 (l)
c) \(M_A=\dfrac{0,6.28+0,7.2}{0,6+0,7}=14\left(g/mol\right)\)
=> \(d_{A/O_2}=\dfrac{14}{32}=0,4375\)