Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a,n_{Na}=\dfrac{m_{Na}}{M_{Na}}=\dfrac{4,6}{23}=0,2\left(mol\right)\\ n_{Cl_2}=\dfrac{V_{Cl_2\left(đktc\right)}}{22,4}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\\ PTHH:2Na+Cl_2\rightarrow2NaCl\\ Vì:\dfrac{0,2}{2}< \dfrac{0,2}{1}\Rightarrow Cl_2dư\\ \Rightarrow n_{Cl_2\left(dư\right)}=0,2-\dfrac{0,2}{2}=0,1\left(mol\right)\\ \Rightarrow m_{Cl_2\left(dư\right)}=0,1.71=7,1\left(g\right)\\ b,n_{NaCl}=n_{Na}=0,2\left(mol\right)\\ \Rightarrow m_{NaCl}=58,5.0,2=11,7\left(g\right)\)

\(n_{H_2}=\dfrac{0,336}{22,4}=0,015(mol)\\ PTHH:Mg+2HCl\to MgCl_2+H_2\\ MgO+2HCl\to MgCl_2+H_2O\\ \Rightarrow n_{Mg}=0,015(mol)\\ \Rightarrow \%_{Mg}=\dfrac{0,015.24}{1,5}.100\%=24\%\\ \Rightarrow \%_{MgO}=100\%-24\%=76\%\)
Chọn A

Mg+2HCl->MgCl2+H2
a..............................a(mol)
Fe+2HCl->FeCl2+H2
b............................b(mol)
=>nCu=3,2/64=0,05mol
=>%mCu=(3,2.100%)/11,2=28,6%
\(=>\left\{{}\begin{matrix}24a+56b=11,2-3,2\\a+b=0,2\end{matrix}\right.=>\left\{{}\begin{matrix}a=0,1\\b=0,1\end{matrix}\right.\)
=>mMg=24.0,1=2,4g=>%Mg=(2,4.100%)/11,2=21,4%
=>%Fe=100%-21,4%-28,6%=50%
b, MgCl2+2NaOH->Mg(OH)2+2NaCL
FeCl2+2NaOH->Fe(OH)2+2NaCl
=>m(kết tủa)=mMg(OH)2+mFe(OH)2
=0,1(58+90)=14,8g
a) mCu= m(k tan)= 3,2(g)
=> m(Mg, Fe)= 11,2- 3,2=8(g)
nH2= 4,48/22,4=0,2(mol)
PTHH: Mg + 2 HCl -> MgCl2 + H2
a______________2a__a______a(mol)
Fe + 2 HCl -> FeCl2 + H2
b____2b_____b_____b(mol)
Ta có hpt:
\(\left\{{}\begin{matrix}24a+56b=8\\a+b=0,2\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,1\\b=0,1\end{matrix}\right.\\ \Leftrightarrow\left\{{}\begin{matrix}m_{Mg}=24.0,1=2,4\left(g\right)\\m_{Fe}=56.0,1=5,6\left(g\right)\end{matrix}\right.\)
=> %mMg= (2,4/11,2).100=21,429%
%mFe= (5,6/11,2).100=50%
=>%mCu= (3,2/11,2).100=28,571%
b/ MgCl2 + 2 NaOH -> Mg(OH)2 + 2 NaCl
0,1___________________0,1(mol)
FeCl2 + 2 NaOH -> Fe(OH)2 +2 NaCl
0,1__________________0,1(mol)
m(kt)=mMg(OH)2 + mFe(OH)2= 58.0,1+ 90.0,1= 14,8(g)

Đặt a,b là số mol Mg, R trong 8 gam A. Đặt x,y là hoá trị thấp cao của R
mA = 24a + bR = 8 (1)
Với HCl -> 2a + bx = 0,2 .2 (2)
Trong 9,6 gam A ( gấp 1,2 lần 8 gam A ) chứa 1,2a và 1,2b mol Mg, R
Với Cl2 -> 2 . 1,2a + 1,2by = 2 ( 30,9 - 9,6 ) / 71 (3)
Với 1 ≤ x ≤ y ≤ 3 -> Chọn x = 2; y = 3
(2)(3) -> a = b = 0,1
(1) -> R= 56 -> = Fe

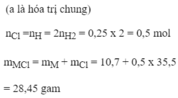
\(n_{Mg}=\dfrac{7,2}{24}=0,3\left(mol\right)\\ n_{Cl_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\\ Mg+Cl_2\rightarrow\left(t^o\right)MgCl_2\\ a,Vì:\dfrac{0,2}{1}< \dfrac{0,3}{1}\Rightarrow Mgdư\\n_{Mg\left(p.ứ\right)}=n_{MgCl_2}=n_{Cl_2}=0,2\left(mol\right)\\ \Rightarrow n_{Mg\left(dư\right)}=0,3-0,2=0,1\left(mol\right)\\ m_{Mg\left(dư\right)}=0,1.24=2,4\left(g\right)\\ b,m_{MgCl_2}=0,2.95=19\left(g\right)\)
\(n_{Mg}=\dfrac{m}{M}=\dfrac{7,2}{24}=0,3\) (mol)
\(n_{Cl_2}=\dfrac{V}{22,4}=\dfrac{4,48}{22,4}=0,2\)(mol)
PTHH : Mg + Cl2 ---> MgCl2
1 : 1 : 1
Dễ thấy : \(\dfrac{n_{Mg}}{1}>\dfrac{n_{Cl_2}}{1}\)
=> Mg dư 0,1 mol
=> \(m_{Mg}=n.M=0,1.24=2,4\left(g\right)\)
=> \(n_{MgCl_2}=0,2\left(mol\right)\) => \(m_{MgCl_2}=n.M=0,2.\left(24+71\right)=19\left(g\right)\)
\(n_{Mg}=\dfrac{m}{M}=\dfrac{7,2}{24}=0,3\) (mol)
\(n_{Cl_2}=\dfrac{V}{22,4}=\dfrac{4,48}{22,4}=0,2\)(mol)
PTHH : Mg + Cl2 ---> MgCl2
1 : 1 : 1
Dễ thấy : \(\dfrac{n_{Mg}}{1}>\dfrac{n_{Cl_2}}{1}\)
=> Mg dư 0,1 mol
=> \(m_{Mg}=n.M=0,1.24=2,4\left(g\right)\)
=> \(n_{MgCl_2}=0,2\left(mol\right)\) => \(m_{MgCl_2}=n.M=0,2.\left(24+71\right)=19\left(g\right)\)