Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

PTHH: \(MgO+2HCl\rightarrow MgCl_2+H_2O\)
Ta có: \(n_{MgO}=\dfrac{10}{40}=0,25\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{HCl}=0,5\left(mol\right)\\n_{MgCl_2}=0,25\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}C_{M_{HCl}}=\dfrac{0,5}{0,5}=1\left(M\right)\\C_{M_{MgCl_2}}=\dfrac{0,25}{0,5}=0,5\left(M\right)\end{matrix}\right.\)
Ta có: nMgO = \(\dfrac{10}{40}=0,25\left(mol\right)\)
a. PTHH: MgO + 2HCl ---> MgCl2 + H2O
Theo PT: nHCl = 2.nMgO = 2.0,25 = 0,5(mol)
Đổi 500ml = 0,5 lít
=> \(C_{M_{HCl}}=\dfrac{0,25}{0,5}=0,5M\)
b. Ta có: \(V_{dd_{MgCl_2}}=10+0,5=10,5\left(lít\right)\)
Theo PT: \(n_{MgCl_2}=n_{MgO}=0,25\left(mol\right)\)
=> \(C_{M_{MgCl_2}}=\dfrac{0,25}{10,5}=0,024M\)

\(2Al + 6HCl \rightarrow 2AlCl_3 + 3H_2\)
\(n_{Al}= \dfrac{5,4}{27}= 0,2 mol\)
Theo PTHH:
\(n_{AlCl_3}= n_{Al}= 0,2 mol\)
\(\Rightarrow m_{AlCl_3}= 0,2 . 133,5=26,7 g\)
Theo PTHH:
\(n_{H_2}= \dfrac{3}{2} n_{Al}= 0,3 mol\)
\(\Rightarrow V= 0,3 . 22,4= 6,72 l\)
b)
Theo PTHH:
\(n_{HCl}= 3n_{Al}= 0,6 mol\)
\(\Rightarrow m_{HCl}= 0,6 . 36,5=21,9 g\)
\(\Rightarrow m_{dd HCl}= \dfrac{21,9 . 100}{15}= 146 g\) ( nếu ở tử là : 21,9 . 100% thì ở mẫu bạn chia cho 15% nhé)

a) \(n_{CH_3COOH}=\dfrac{100.12\%}{60}=0,2\left(mol\right)\)
PTHH: CH3COOH + NaHCO3 --> CH3COONa + CO2 + H2O
0,2-------->0,2----------->0,2--------->0,2
=> mNaHCO3 = 0,2.84 = 16,8 (g)
=> \(m_{dd.NaHCO_3}=\dfrac{16,8.100}{8,4}=200\left(g\right)\)
b) mCH3COONa = 0,2.82 = 16,4 (g)
mdd sau pư = 100 + 200 - 0,2.44 = 291,2 (g)
=> \(C\%_{dd.muối}=\dfrac{16,4}{291,2}.100\%=5,632\%\)

\(m_{ct}=\dfrac{12.100}{100}=12\left(g\right)\)
\(n_{CH3COOH}=\dfrac{12}{60}=0,2\left(mol\right)\)
Pt : \(CH_3COOH+NaHCO_3\rightarrow CH_3COONa+CO_2+H_2O|\)
1 1 1 1 1
0,2 0,2 0,2 0,2
a) \(n_{NaHCO3}=\dfrac{0,2.1}{1}=0,2\left(mol\right)\)
⇒ \(m_{NaHCO3}=0,2.84=16,8\left(g\right)\)
\(m_{ddNaHCO3}=\dfrac{16,8.100}{8,4}=200\left(g\right)\)
b) \(n_{CH3COONa}=\dfrac{0,2.1}{1}=0,2\left(mol\right)\)
⇒ \(m_{CH3COONa}=0,2.82=16,4\left(g\right)\)
\(m_{ddspu}=100+200-\left(0,2.44\right)=291,2\left(g\right)\)
\(C_{CH3COONa}=\dfrac{16,4.100}{291,2}=5,63\)0/0
Chúc bạn học tốt

\(n_{Zn}=0,2\left(mol\right)\\ a,Zn+2HCl\rightarrow ZnCl_2+H_2\\ b,n_{H_2}=n_{ZnCl_2}=n_{Zn}=0,2\left(mol\right);n_{HCl}=2.0,2:0,9=\dfrac{4}{9}\left(mol\right)\\ V_{H_2\left(đktc\right)}=0,2.22,4=4,48\left(l\right)\\ c,C_{MddHCl}=\dfrac{\dfrac{4}{9}}{0,2}=\dfrac{4}{45}\left(M\right)\\ m_{ddHCl}=\dfrac{\dfrac{4}{9}.36,5.100}{14,6}=111,111\left(g\right)\\ d,Zn+H_2SO_4\rightarrow ZnSO_4+H_2\\ n_{H_2SO_4}=n_{Zn}=0,2\left(mol\right)\\ m_{ddaxit}=\dfrac{0,2.98.100}{19,6}=100\left(g\right)\\ V_{ddH_2SO_4}=\dfrac{100}{1,14}=87,719\left(ml\right)\)

a) PTHH: \(Na_2SO_3+H_2SO_4\rightarrow Na_2SO_4+H_2O+SO_2\uparrow\)
Ta có: \(n_{Na_2SO_3}=\dfrac{12,6}{126}=0,1\left(mol\right)=n_{SO_2}\) \(\Rightarrow V_{SO_2}=0,1\cdot22,4=2,24\left(l\right)\)
b) Ta có: \(\left\{{}\begin{matrix}n_{SO_2}=0,1\left(mol\right)\\n_{Ca\left(OH\right)_2}=1,4\cdot0,1=0,14\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\) Tạo muối trung hòa
PTHH: \(Ca\left(OH\right)_2+SO_2\rightarrow CaSO_3\downarrow+H_2O\)
Ta có: \(\left\{{}\begin{matrix}n_{CaSO_3}=0,1\left(mol\right)=n_{Ns_2SO_4}\\n_{Ca\left(OH\right)_2\left(dư\right)}=0,04\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}m_{CaSO_3}=0,1\cdot120=12\left(g\right)\\m_{Na_2SO_4}=0,1\cdot142=14,2\left(g\right)\\m_{Ca\left(OH\right)_2\left(dư\right)}=0,04\cdot74=2,96\left(g\right)\end{matrix}\right.\)
\(n_{Na_2SO_3}=0,1\left(mol\right)\\ PTHH:Na_2SO_3+H_2SO_4\rightarrow Na_2SO_4+H_2O+SO_2\)
(mol) 0,1 0,1 0,1 0,1
\(a.V_{SO_2}=0,1.22,4=2,24\left(l\right)\)
\(b.n_{Ca\left(OH\right)_2}=0,14\left(mol\right)\)
Do \(\dfrac{n_{OH}}{n_{SO_2}}=\dfrac{0,28}{0,1}=2.8>2\rightarrow\) Tạo muối trung hòa và Ca(OH)2 dư 0,04(mol)
\(PTHH:Ca\left(OH\right)_2+SO_2\rightarrow CaSO_3+H_2O\)
(mol) 0,1 0,1 0,1 0,1
\(m_{Ca\left(OH\right)_2\left(du\right)}=0,04.74=2,96\left(g\right)\\ m_{CaSO_3}=12\left(g\right)\\ m_{H_2O}=0,1.18=1,8\left(g\right)\)
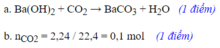
nZn=6,5/65=0,1mol
đổi 500ml = 0,5l
nHCl = 0,5.1 =0,5mol
pt : Zn + 2HCl ------> ZnCl2 + H2
n có: 0,1 0,5
n pứ: 0,1 ----->0,2----------->0,1-------->0,1
n dư: 0 0,3
VH2= 0,1.22.4 =2,24l
mZnCl2= 0,1.136=13,6g
Vdd sau pứ =VddHCl =0,5l
CM(HCl dư) = 0,3/0,5=0,6M
CM(ZnCl2) = 0,1/0,5 =0,2M
a)
nZn = \(\dfrac{6,5}{65}=0,1mol\)
500ml = 0,5 (l)
Ta có: CM = \(\dfrac{n}{V}=\dfrac{n}{0,5}=1\Rightarrow n_{HCl}=0,5mol\)
Zn + 2HCl -----> ZnCl2 + H2
0,1 0,5
Xét: \(\dfrac{0,1}{1}< \dfrac{0,5}{2}\Rightarrow\) Zn hết, HCl dư
\(\Rightarrow\) \(n_{H_2}=0,1mol\) \(\Rightarrow V_{H_2}=0,1.22,4=2,24l\)
b) \(n_{ZnCl_2}=0,1mol\)
\(\Rightarrow\)\(m_{ZnCl_2}=0,1.136=13,6g\)
c) CM = \(\dfrac{n_{ZnCl_2}}{V}\) = \(\dfrac{0,1}{0,5}\)= 0,2M
nHCl dư = 0,5 - 0,2 = 0,3 mol
CM = \(\dfrac{n_{HCL}}{V}\) = \(\dfrac{0,3}{0,5}\) = 0,6M