Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) PTHH: 2Al + 3H2SO4 --> Al2(SO4)3 + 3H2
b) \(n_{H_2}=\dfrac{0,672}{22,4}=0,03\left(mol\right)\)
PTHH: 2Al + 3H2SO4 --> Al2(SO4)3 + 3H2
_____0,02<---0,03<---------------------0,03
=> \(\left\{{}\begin{matrix}\%Al=\dfrac{0,02.27}{2,16}.100\%=25\%\\\%Cu=100\%-25\%=75\%\end{matrix}\right.\)
c) mH2SO4 = 0,03.98 = 2,94 (g)
=> \(C\%\left(H_2SO_4\right)=\dfrac{2,94}{200}.100\%=1,47\%\)

PTHH: \(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\)
Ta có: \(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)=n_{Fe}\)
\(\Rightarrow\left\{{}\begin{matrix}n_{HCl}=0,2\left(mol\right)\\\%m_{Fe}=\dfrac{0,1\cdot56}{12}\cdot100\%\approx46,67\%\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}C_{M_{HCl}}=\dfrac{0,2}{0,2}=1\left(M\right)\\\%m_{Cu}=53,33\%\end{matrix}\right.\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
Cu không phản ứng
\(nH_2=nFe=\dfrac{2,24}{22,4}=0,1mol\)
\(\rightarrow mFe=0,1.56=5,6gam\)
\(\rightarrow\%mFe=\dfrac{5,6}{12}.100\%=46,\left(6\right)\%\)
\(\rightarrow\%mCu=100\%-46,\left(6\right)\%=53,\left(3\right)\%\)
c)
\(CM_{HCl}=\dfrac{0,1.2}{0,2}=1M\)

PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
Ta có: \(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{Zn}=0,3\left(mol\right)=n_{ZnCl_2}\\n_{HCl}=0,6\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{0.3\cdot65}{35,5}\cdot100\%\approx54,93\%\\\%m_{Cu}=45,07\%\\C\%_{HCl}=\dfrac{0,6\cdot36,5}{500}\cdot100\%=4,38\%\\m_{ZnCl_2}=0,3\cdot136=40,8\left(g\right)\end{matrix}\right.\)
Mặt khác: \(\left\{{}\begin{matrix}m_{Cu}=35,5-0,3\cdot65=16\left(g\right)\\m_{H_2}=0,3\cdot2=0,6\left(g\right)\end{matrix}\right.\)
\(\Rightarrow m_{dd}=m_{KL}+m_{ddHCl}-m_{Cu}-m_{H_2}=518,9\left(g\right)\)
\(\Rightarrow C\%_{ZnCl_2}=\dfrac{40,8}{518,9}\cdot100\%\approx7,86\%\)

\(a.n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\\ Đặt:\left\{{}\begin{matrix}n_{Al}=a\left(mol\right)\\n_{Mg}=b\left(mol\right)\end{matrix}\right.\left(a,b>0\right)\\ 2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\\ Mg+H_2SO_4\rightarrow MgSO_4+H_2\\ \rightarrow\left\{{}\begin{matrix}27a+24b=5,1\\1,5a+b=0,25\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,1\\b=0,1\end{matrix}\right.\\ \left\{{}\begin{matrix}\%m_{Al}=\dfrac{27.0,1}{5,1}.100\approx52,941\%\\\%m_{Mg}\approx47,059\%\end{matrix}\right.\)
\(b.m_{ddH_2SO_4}=\dfrac{0,25.98.100}{9,8}=250\left(g\right)\\ m_{ddsau}=m_{Al,Mg}+m_{ddH_2SO_4}-m_{H_2}=5,1+250-0,25.2=254,6\left(g\right)\\ C\%_{ddAl_2\left(SO_4\right)_3}=\dfrac{0,05.342}{254,6}.100\approx6,716\%\\ C\%_{ddMgSO_4}=\dfrac{0,1.120}{254,6}.100\approx4,713\%\)

\(m_{ct}=\dfrac{10.159}{100}=15,9\left(g\right)\)
\(n_{Na2CO3}=\dfrac{15,9}{106}=0,15\left(mol\right)\)
Pt : \(MgCl_2+Na_2CO_3\rightarrow MgCO_3+2NaCl|\)
1 1 1 2
a 1a 0,2
\(CaCl_2+Na_2CO_3\rightarrow CaCO_3+2NaCl|\)
1 1 1 2
b 1b 0,1
a) Gọi a là số mol của MgCl2
b là số mol của CaCl2
\(m_{MgCl2}+m_{CaCl2}=15,05\left(g\right)\)
⇒ \(n_{MgCl2}.M_{MgCl2}+n_{CaCl2}.M_{CaCl2}=15,05g\)
⇒ 95a + 111b = 15,05g(1)
Ta có : 1a + 1b = 0,15(2)
Từ(1),(2), ta có hệ phương trình :
95a + 111b = 15,05g
1a + 1b = 0,15
⇒ \(\left\{{}\begin{matrix}a=0,1\\b=0,05\end{matrix}\right.\)
\(m_{MgCl2}=0,1.95=9,5\left(g\right)\)
\(m_{CaCl2}=0,05.111=5,55\left(g\right)\)
0/0MgCl2 = \(\dfrac{9,5.100}{15,05}=63,12\)0/0
0/0CaCl2 = \(\dfrac{5,55.100}{15,05}=36,88\)0/0
b) \(n_{NaCl\left(tổng\right)}=0,2+0,1=0,3\left(mol\right)\)
⇒ \(m_{NaCl}=0,3.58,5=17,55\left(g\right)\)
\(m_{ddspu}=159+141=300\left(g\right)\)
\(C_{NaCl}=\dfrac{17,55.100}{300}=5,85\)0/0
Chúc bạn học tốt

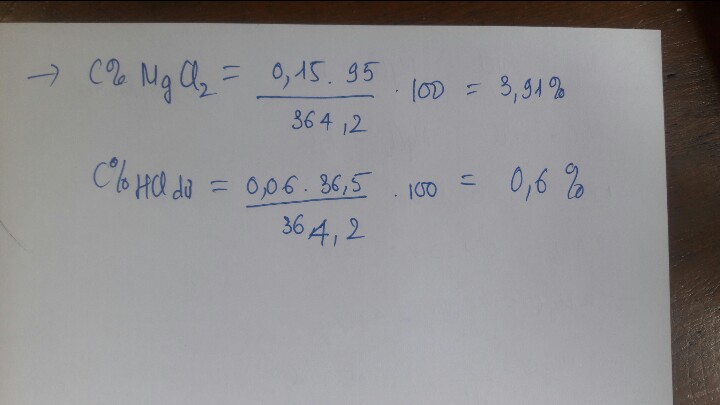
\(n_{CO_2}=\dfrac{2,24}{22,4}=0,1mol\)
a)\(CaCO_3+2HCl\rightarrow CaCl_2+H_2O+CO_2\)
0,1 0,2 0,1 0,1 0,1
b)\(m_{HCl}=0,1\cdot36,5=3,65\left(g\right)\)
\(a\%=\dfrac{3,65}{100}\cdot100\%=3,65\%\)
c)\(m_{CaCO_3}=0,1\cdot100=10\left(G\right)\)
\(\Rightarrow\%m_{CaCO_3}=\dfrac{10}{16}\cdot100\%=62,5\%\)
\(\Rightarrow\%m_{CaCl_2}=100\%-62,5\%=37,5\%\)
d)\(m_{CaCl_2}=0,1\cdot111=11,1\left(g\right)\)
\(m_{H_2O}=0,1\cdot18=1,8\left(g\right)\)
\(m_{ddsau}=10+100-0,1\cdot44-1,8=103,8\left(g\right)\)
\(\Rightarrow C\%=\dfrac{11,1}{103,8}\cdot100\%=10,7\%\)