Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a,2KMnO_4+16HCl_{đặc}\rightarrow\left(t^o\right)2KCl+2MnCl_2+5Cl_2+8H_2O\\ 2Fe+3Cl_2\rightarrow\left(t^o\right)2FeCl_3\\ Ta.có:n_{FeCl_3}=\dfrac{39}{162,5}=0,24\left(mol\right)\\ n_{Fe}=n_{FeCl_3}=0,24\left(mol\right)\\ n_{Cl_2}=\dfrac{3}{2}.0,24=0,36\left(mol\right)\\ n_{K_2MnO_4}=\dfrac{2}{5}.0,36=0,144\left(mol\right)\\ n_{HCl}=\dfrac{16}{5}.0.36=1,152\left(mol\right)\\ \Rightarrow a=m_{KMnO_4}=0,144.158=22,752\left(g\right)\\ b=C_{MddHCl}=\dfrac{1,152}{0,1}=11,52\left(M\right)\\ x=m_{Fe}=0,24.56=13,44\left(g\right)\\ V=V_{Cl_2\left(đktc\right)}=0,36.22,4=8,064\left(l\right) \)
\(b,n_{KCl}=n_{MnCl_2}=\dfrac{2}{5}.0,36=0,144\left(mol\right)\\ KCl+AgNO_3\rightarrow AgCl\downarrow\left(trắng\right)+KNO_3\\ MnCl_2+2AgNO_3\rightarrow2AgCl\downarrow\left(trắng\right)+Mn\left(NO_3\right)_2\\ n_{AgNO_3}=n_{AgCl}=n_{KCl}+2.n_{MnCl_2}=0,144+2.0,144=0,432\left(mol\right)\\ \Rightarrow m_{AgCl\downarrow\left(trắng\right)}=143,5.0,432=61,992\left(g\right)\\ m_{AgNO_3}=0,432.170=73,44\left(g\right)\\ \Rightarrow m_{ddAgNO_3}=\dfrac{73,44.100}{5}=1468,8\left(g\right)\)

Cl2+2NaBr ->2NaCl+Br2
0,04 -0,08-------0,08-----0,04 mol
Cl2+2NaI ->2NaCl+I2
0,03--0,06---0,06-----0,03 mol
n NaBr =0,8.0,1=0,08 mol
n NaI=0,6.0,1=0,06 mol
=>V Cl2=(0,04+0,03).22,4=1,568l
=>m Br2=0,04 .160=6,4g
=>m I2=0,03.254=7,62g
b)
NaCl+AgNO3->AgCl+NaNO3
0,14-----------------0,14 mol
=>m AgCl=0,14.143,5=20,09g
c)2KMnO4+16HCl->2KCl+2MnCl2+5Cl2+8H2O
0,028--------------------------------------0,07 mol
=>m KMnO4=0,028.158=4,424g

a) Gọi x,y lần lượt là số mol Fe, Cu trong hhX (x,y>0) (mol)
- Khi cho X t/d hoàn toàn với khí Clo dư:
\(2Fe+3Cl_2\rightarrow\left(t^o\right)2FeCl_3\\ Cu+Cl_2\rightarrow\left(t^o\right)CuCl_2\\ \Rightarrow162,5x+135y=59,5\left(1\right)\)
- Khi cho X tác dụng hoàn toàn với dd HCl 36,5%. Cu sẽ không tác dụng mà chỉ có Fe tham gia phản ứng.
\(Fe+2HCl\rightarrow FeCl_2+H_2\\ m_{FeCl_2}=127x=25,4\left(g\right)\left(2\right)\\ \left(1\right),\left(2\right)\Rightarrow\left\{{}\begin{matrix}162,5x+135y=59,5\\127x=25,4\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}x=0,2\\y=0,2\end{matrix}\right.\\ \Rightarrow a=m_{hhX}=m_{Fe}+m_{Cu}=64x+56y=64.0,2+56.0,2=24\left(g\right)\)
Tính phần trăm mỗi muối sau phản ứng chắc ở phản ứng với Clo dư.
\(\%m_{FeCl_3}=\dfrac{0,2.162,5}{0,2.162,5+0,2.135}.100\approx54,622\%\\ \Rightarrow\%m_{CuCl_2}\approx45,378\%\)
b)
\(n_{HCl}=2x=2.0,2=0,4\left(mol\right)\\ \Rightarrow m_{HCl}=0,4.36,5=14,6\left(g\right)\\ \Rightarrow m_{ddHCl}=\dfrac{14,6.100}{36,5}=40\left(g\right)\\ \Rightarrow V_{ddHCl}=\dfrac{m_{ddHCl}}{D_{ddHCl}}=\dfrac{40}{1,25}=32\left(ml\right)=0,032\left(l\right)\)

\(Đặt:n_{MnO_2}=a\left(mol\right),n_{KMnO_4}=b\left(mol\right)\)
\(m_{hh}=87a+158b=37.96\left(g\right)\left(1\right)\)
\(n_{Cl_2}=\dfrac{10.08}{22.4}=0.45\left(mol\right)\)
\(2KMnO_4+16HCl\rightarrow2KCl+2MnCl_2+5Cl_2+8H_2O\)
\(MnO_2+4HCl\rightarrow MnCl_2+Cl_2+2H_2O\)
\(n_{Cl_2}=a+2.5b=0.45\left(mol\right)\left(2\right)\)
\(\left(1\right),\left(2\right):a=0.4,b=0.02\)
\(\%MnO_2=\dfrac{0.4\cdot87}{37.96}\cdot100\%=91.68\%\\\%KMnO_4=100-91.68=8.32\% \)
\(m_M=m_{KCl}+m_{MnCl_2}=0.02\cdot74.5+\left(0.4+0.02\right)\cdot126=54.41g\)

n chất rắn =m Cu=19,2 g
=>m Mg, Al=29,4-19,2=10,2g
Mg+2HCl->MgCl2+H2
x-------------------------x
2Al+6HCl->2AlCl3+3H2
y--------------------------\(\dfrac{3}{2}\)y
=>Ta có :
\(\left\{{}\begin{matrix}24x+27y=10,2\\x+\dfrac{3}{2}y=\dfrac{11,2}{22,4}\end{matrix}\right.\)
=>x=0,2 mol , y=0,2 mol
=>% Cu=\(\dfrac{19,2}{29,4}\).100=65,3%
=>%Mg=\(\dfrac{0,2.24}{29,4}\).100=16,32%
=>%Al=100-65,3-16,32=18,28%
b)MgCl2+2AgNO3->2AgCl+Mg(NO3)2
0,2----------------------0,4
AlCl3+3AgNO3->Al(NO3)3+3AgCl
0,2-----------------------------------------0,6
=>m AgCl=(0,6+0,4).143,5=143,5g


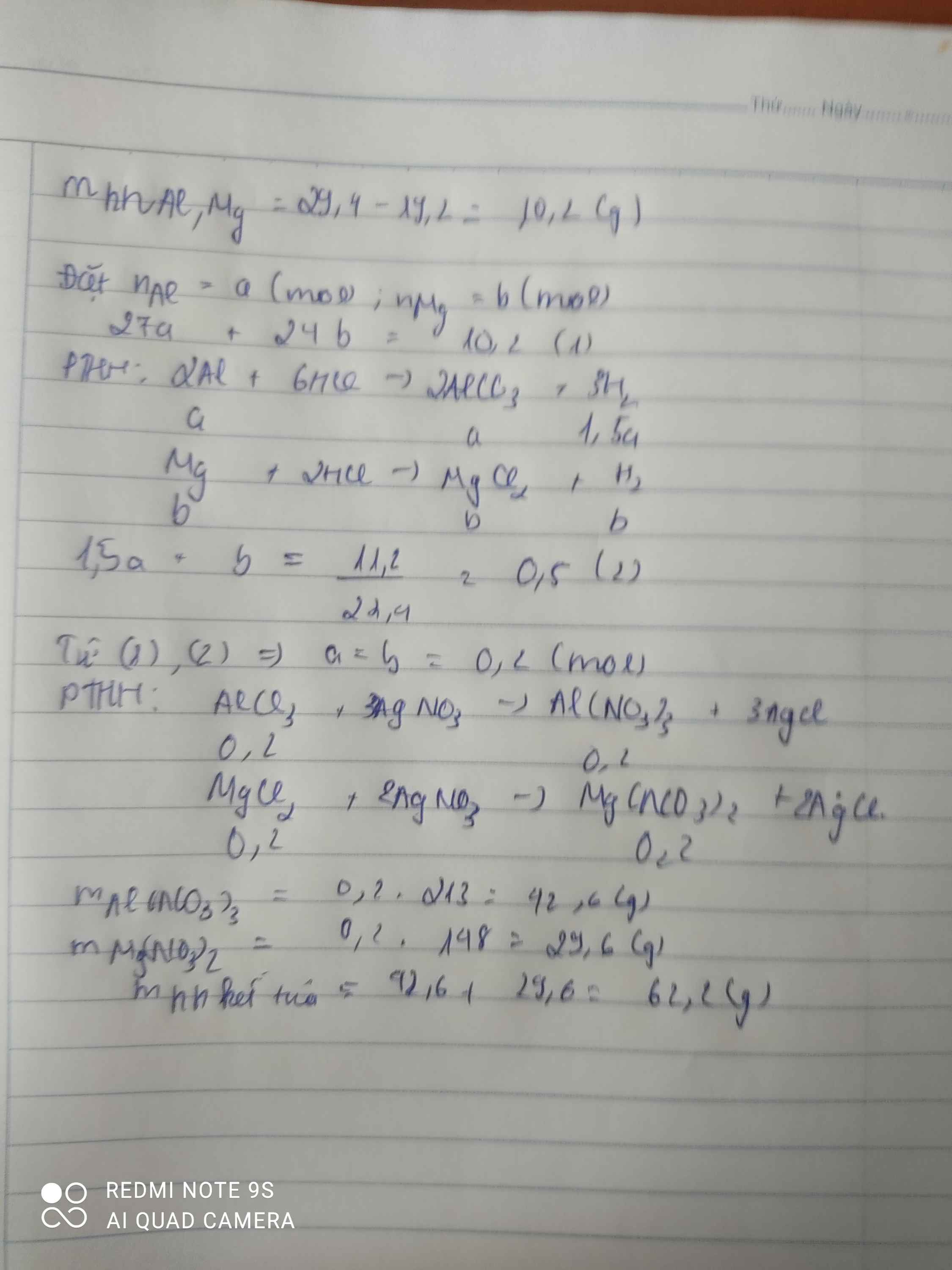
\(a,n_{Cl_2}=\dfrac{15,62}{71}=0,22\left(mol\right)\\ Đặt:n_{Fe}=a\left(mol\right);n_{Cu}=b\left(mol\right)\left(a,b>0\right)\\ 2Fe+3Cl_2\rightarrow\left(t^o\right)2FeCl_3\\ Cu+Cl_2\rightarrow\left(t^o\right)CuCl_2\\ \Rightarrow\left\{{}\begin{matrix}56a+64b=10,88\\1,5a+b=0,22\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,08\\b=0,1\end{matrix}\right.\\ \Rightarrow\%m_{Fe}=\dfrac{0,08.56}{10,88}.100\approx41,176\%\Rightarrow\%m_{Cu}\approx58,824\%\\ b,FeCl_3+3AgNO_3\rightarrow Fe\left(NO_3\right)_3+3AgCl\downarrow\\ CuCl_2+2AgNO_3\rightarrow Cu\left(NO_3\right)_2+2AgCl\\ n_{AgNO_3}=3.0,08+2.0,1=0,44\left(mol\right)\\ m_{AgNO_3}=0,24.170=40,8\left(g\right)\\ m_{ddAgNO_3}=\dfrac{40,8}{24\%}=170\left(g\right)\)
\(c,2NaI+Cl_2\rightarrow2NaCl+I_2\\ n_{NaI}=2.n_{Cl_2}=2.0,22=0,44\left(mol\right)\Rightarrow a=C_{MddNaI}=\dfrac{0,44}{0,1}=4,4\left(M\right)\\ n_{I_2}=n_{Cl_2}=0,22\left(mol\right)\Rightarrow m_{I_2}=254.0,22=55,88\left(g\right)\)
\(d,MnO_2+4HCl_{\left(đặc\right)}\rightarrow\left(t^o\right)MnCl_2+Cl_2+2H_2O\\ n_{MnO_2}=n_{Cl_2}=0,22\left(mol\right)\\ \Rightarrow x=m_{MnO_2}=87.0,22=19,14\left(g\right)\\ n_{HCl}=4.0,22=0,88\left(mol\right)\\ y=C_{MddHCl}=\dfrac{0,88}{0,05}=17,6\left(M\right)\)