Đốt cháy hoàn toàn rượu etylic cần dùng 4,48 lít oxi(đktc) .a/ Tính thể tích chất khí sinh ra ở đktc .b/Tính khối lượng etylic tham gia phản ứng biết hiệu suất phản ứng là 90%
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


\(a.C_2H_5OH+3O_2-^{t^o}\rightarrow2CO_2+3H_2O\\ n_{C_2H_5OH}=0,3\left(mol\right)\\ n_{CO_2}=2n_{C_2H_5OH}=0,6\left(mol\right)\\ \Rightarrow V_{CO_2}=0,6.22,4=13,44\left(l\right)\\ b.n_{O_2}=3n_{C_2H_5OH}=0,6\left(mol\right)\\ MàV_{O_2}=\dfrac{1}{5}V_{kk}\\ \Rightarrow V_{kk}=V_{O_2}.5=0,6.22,4.5=67,2\left(l\right)\\ c.n_{NaOH}=0,9\left(mol\right)\\ Tacó:\dfrac{n_{NaOH}}{n_{CO_2}}=\dfrac{0,9}{0,6}=1,5\\ \Rightarrow Tạora2muốiNaHCO_3vàNa_2CO_3\\ Đặt:n_{NaHCO_3}=x\left(mol\right);n_{Na_2CO_3}=y\left(mol\right)\\ \Rightarrow\left\{{}\begin{matrix}x+y=0,6\left(BTnguyento\left(C\right)\right)\\x+2y=0,9\left(BTnguyento\left(Na\right)\right)\end{matrix}\right.\\ \Rightarrow\left\{{}\begin{matrix}x=0,3\\y=0,3\end{matrix}\right.\\ \Rightarrow m_{muối}=0,3.84+0,3.106=57\left(g\right)\)

a) C2H5OH + 3O2 --to--> 2CO2 + 3H2O
b) \(n_{C_2H_5OH}=\dfrac{46}{46}=1\left(mol\right)\)
PTHH: C2H5OH + 3O2 --to--> 2CO2 + 3H2O
1----->3----------->2------->3
=> VO2 = 22,4.3 = 67,2 (l)
c) mH2O = 3.18 = 54 (g)
d) VCO2 = 2.22,4 = 44,8 (l)

\(n_{CO2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
\(C_2H_5OH+3O_2\xrightarrow[]{t^o}2CO_2+3H_2O\)
0,1 0,3 0,2
b) \(m_{C2H5OH}=0,1.46=4,6\left(g\right)\)
c) \(V_{O2\left(dktc\right)}=0,3.22,4=6,72\left(l\right)\)
Chúc bạn học tốt

\(n_{CO_2}=\dfrac{4,48}{22,4}=0,2mol\)
\(C_2H_5OH+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
0,2 0,6 0,4 0,6
a)\(m_{C_2H_5OH}=0,2\cdot46=9,2g\)
b)\(V_{O_2}=0,6\cdot22,4=13,44l\)
\(\Rightarrow V_{kk}=5V_{O_2}=5\cdot13,44=67,2l\)

a, \(C_2H_6O+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
b, \(n_{CO_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
Theo PT: \(n_{O_2}=\dfrac{3}{2}n_{CO_2}=0,3\left(mol\right)\)
\(\Rightarrow V_{O_2}=0,3.22,4=6,72\left(l\right)\)
c, \(n_{C_2H_6O}=\dfrac{1}{2}n_{CO_2}=0,1\left(mol\right)\)
\(\Rightarrow m_{C_2H_6O}=0,1.46=4,6\left(g\right)\)
\(\Rightarrow V_{C_2H_6O}=\dfrac{4,6}{0,8}=5,75\left(ml\right)\)
Độ rượu = \(\dfrac{5,75}{50}.100=11,5^o\)

a) \(n_{O_2}=\dfrac{11,2.20\%}{22,4}=0,1\left(mol\right)\)
PTHH: 2Mg + O2 --to--> 2MgO
0,2<--0,1--------->0,2
=> mMg = 0,2.24 = 4,8 (g)
b) nMgO = 0,2.40 = 8 (g)

a, \(C_2H_6O+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
b, \(n_{C_2H_6O}=\dfrac{23}{46}=0,5\left(mol\right)\)
Theo PT: \(n_{O_2}=3n_{C_2H_6O}=1,5\left(mol\right)\)
\(\Rightarrow V_{O_2}=1,5.22,4=33,6\left(l\right)\)
c, \(V_{C_2H_6O}=\dfrac{100.46}{100}=46\left(ml\right)\)
\(\Rightarrow m_{C_2H_6O}=46.0,8=36,8\left(g\right)\)
\(\Rightarrow n_{C_2H_6O}=\dfrac{36,8}{46}=0,8\left(mol\right)\)
PT: \(C_2H_5OH+Na\rightarrow C_2H_5ONa+\dfrac{1}{2}H_2\)
Theo PT: \(n_{H_2}=\dfrac{1}{2}n_{C_2H_5ONa}=0,4\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,4.22,4=8,96\left(l\right)\)
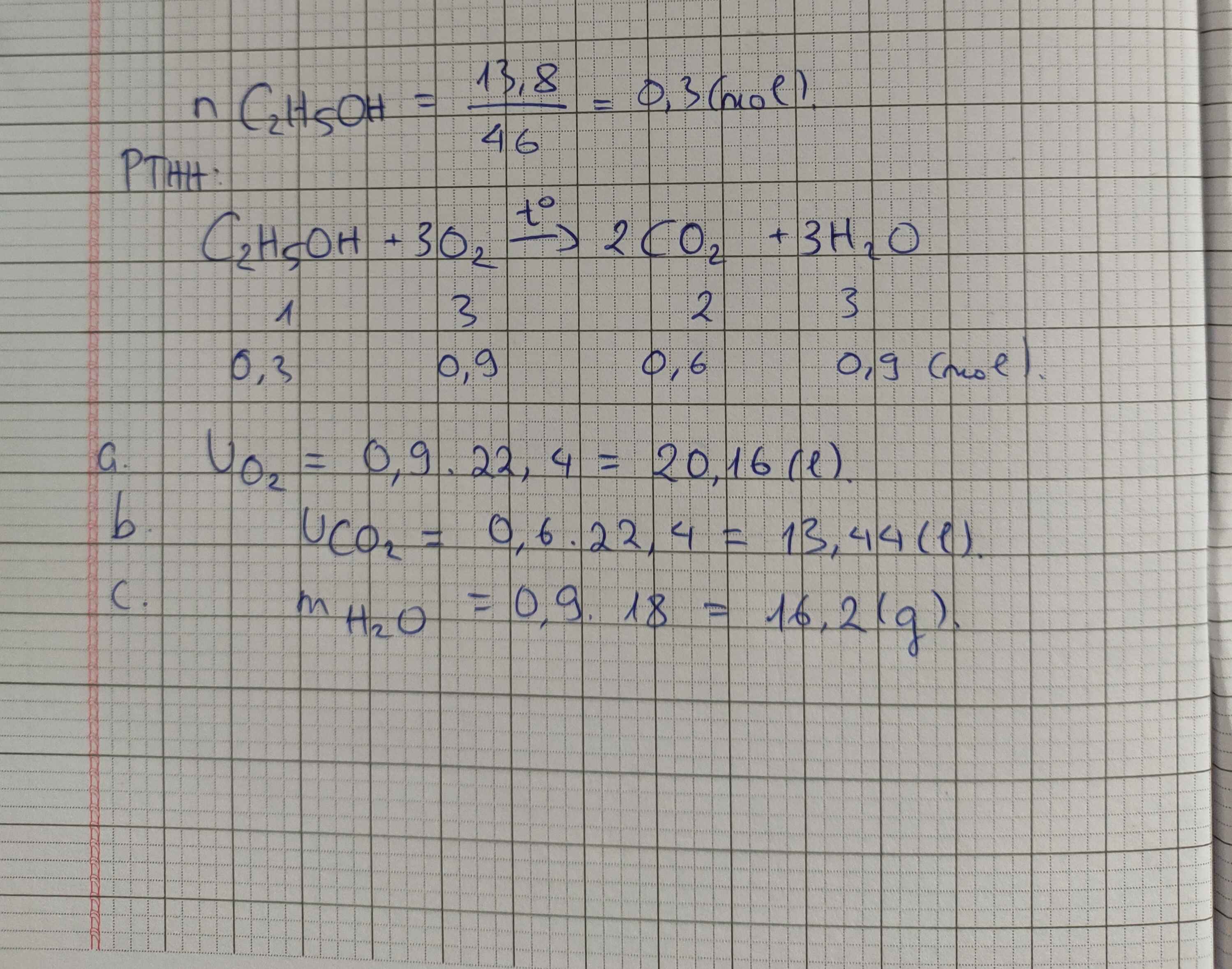
a, \(n_{O_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
PT: \(C_2H_6O+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
Theo PT: \(n_{CO_2}=\dfrac{2}{3}n_{O_2}=\dfrac{2}{15}\left(mol\right)\Rightarrow V_{CO_2}=\dfrac{2}{15}.22,4=\dfrac{224}{75}\left(l\right)\)
b, \(n_{C_2H_6O\left(LT\right)}=\dfrac{1}{3}n_{O_2}=\dfrac{1}{15}\left(mol\right)\)
Mà: H = 90%
\(\Rightarrow n_{C_2H_6O\left(TT\right)}=\dfrac{\dfrac{1}{15}}{90\%}=\dfrac{2}{27}\left(mol\right)\)
\(\Rightarrow m_{C_2H_6O}=\dfrac{2}{27}.46=\dfrac{92}{27}\left(g\right)\)