Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(\left\{{}\begin{matrix}n_{H_2SO_4}=0,2.0,5=0,1\left(mol\right)\\n_{MgSO_4}=0,3.0,5=0,15\left(mol\right)\end{matrix}\right.\)
\(n_{MgO}=\dfrac{4}{40}=0,1\left(mol\right)\)
PTHH: 2NaOH + H2SO4 --> Na2SO4 + 2H2O
0,2<-----0,1
2NaOH + MgSO4 --> Mg(OH)2 + Na2SO4
0,2<------------------0,1
Mg(OH)2 --to--> MgO + H2O
0,1<------------0,1
=> nNaOH = 0,2 + 0,2 = 0,4 (mol)
=> mNaOH = 0,4.40 = 16 (g)
=> \(m_1=\dfrac{16.100}{10}=160\left(g\right)\)
m2 = 0,1.58 = 5,8 (g)

\(n_{NaOH}=\dfrac{200.15\%}{40}=0,75\left(mol\right)\)
\(\left\{{}\begin{matrix}n_{H_2SO_4}=0,0001V\left(mol\right)\\n_{Fe_2\left(SO_4\right)_3}=0,00005V\left(mol\right)\end{matrix}\right.\)
PTHH: 2NaOH + H2SO4 --> Na2SO4 + 2H2O
0,0002V<-0,0001V
6NaOH + Fe2(SO4)3 --> 3Na2SO4 + 2Fe(OH)3
0,0003V<-0,00005V---------------->0,0001V
=> 0,0002V + 0,0003V = 0,75
=> V = 1500 (ml)
nFe(OH)3 = 0,15 (mol)
=> m1 = 0,15.107 = 16,05 (g)
PTHH: 2Fe(OH)3 --to--> Fe2O3 + 3H2O
0,15--------->0,075
=> mFe2O3 = 0,075.160 = 12 (g)

\(n_{CuSO_4}=\dfrac{200.16}{160.100}=0,2mol\)
\(n_{NaOH}=\dfrac{200.10}{40.100}=0,5mol\)
CuSO4+2NaOH\(\rightarrow\)Cu(OH)2\(\downarrow\)+Na2SO4
-Ta có tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,5}{2}\rightarrow\)CuSO4 hết, NaOH dư.
Cu(OH)2\(\overset{t^0}{\rightarrow}\)CuO+H2O
\(n_{CuO}=n_{Cu\left(OH\right)_2}=n_{CuSO_4}=0,2mol\)
a=\(m_{CuO}=0,2.80=16gam\)
\(m_{Cu\left(OH\right)_2}=0,2.98=19,6gam\)
\(n_{NaOH\left(pu\right)}=2n_{CuSO_4}=0,4mol\rightarrow n_{NaOH\left(dư\right)}=0,5-0,4=0,1mol\)
\(m_{NaOH\left(dư\right)}=0,1.40=4gam\)
\(n_{Na_2SO_4}=n_{CuSO_4}=0,2mol\rightarrow m_{Na_2SO_4}=0,2.136=27,2gam\)
\(m_{dd}=200+200-19,6=380,4gam\)
C%NaOH=\(\dfrac{4.100}{380,4}\approx1,05\%\)
C%Na2SO4=\(\dfrac{27,2.100}{380,4}\approx7,15\%\)

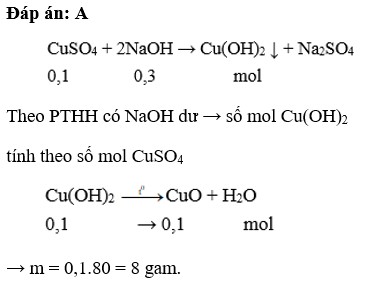
\(CuSO_4+2NaOH->Cu\left(OH\right)_2+Na_2SO_4\)
bđ 0,1 0,25
pư 0,1............0,2...............0,1
spu 0...............0,05.............0,1
\(Cu\left(OH\right)_2-^{t^o}>CuO+H_2O\)
0,1.........................0,1
m CuO = 0,1.(64+16)=8g


nH2SO4=0,2.0,5=0,1(mol)
nMgSO4=0,3.0,5=0,15(mol)
nMgO=\(\dfrac{4}{40}=0,1mol\)
PTHH: 2NaOH + H2SO4 --> Na2SO4 + 2H2O
0,2<-----0,1
2NaOH + MgSO4 --> Mg(OH)2 + Na2SO4
0,2<------------------0,1
Mg(OH)2 --to--> MgO + H2O
0,1<------------0,1
=> nNaOH = 0,2 + 0,2 = 0,4 (mol)
=> mNaOH = 0,4.40 = 16 (g)
=> m1=\(\dfrac{16.100}{10}=160g\)
m2 = 0,1.58 = 5,8 (g)