Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
b, \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
Theo PT: \(n_{H_2}=n_{Zn}=0,1\left(mol\right)\Rightarrow V_{H_2}=0,1.22,4=2,24\left(l\right)\)
c, \(n_{CuO}=\dfrac{12}{80}=0,15\left(mol\right)\)
PT: \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
Xét tỉ lệ: \(\dfrac{0,15}{1}>\dfrac{0,1}{1}\), ta được CuO dư.
Theo PT: \(n_{Cu}=n_{H_2}=0,1\left(mol\right)\Rightarrow m_{Cu}=0,1.64=6,4\left(g\right)\)

a, \(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\)
b, \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
Theo PT: \(n_{H_2}=n_{Zn}=0,2\left(mol\right)\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c, \(n_{Fe_3O_4}=\dfrac{18,56}{232}=0,08\left(mol\right)\)
PT: \(Fe_3O_4+4H_2\underrightarrow{t^o}3Fe+4H_2O\)
Xét tỉ lệ: \(\dfrac{0,08}{1}>\dfrac{0,2}{4}\), ta được Fe3O4 dư.
Theo PT: \(n_{Fe}=\dfrac{3}{4}n_{H_2}=0,15\left(mol\right)\Rightarrow m_{Fe}=0,15.56=8,4\left(g\right)\)

a) 2Al+6HCl→2AlCl3+3H22Al+6HCl→2AlCl3+3H2
b) nAl=5,427=0,2(mol)nAl=5,427=0,2(mol)
Theo phương trình : nH2=32nAl=0,3(mol)nH2=32nAl=0,3(mol)
→VH2(đktc)=0,3.22,4=6,72(l)→VH2(đktc)=0,3.22,4=6,72(l)
c) Chất rắn : 0,2(mol)0,2(mol)
CuO dư : 0,2(mol)Cu0,2(mol)Cu
%CuO=0,2.80(0,2.80+0,2.64).100=55,56%%CuO=0,2.80(0,2.80+0,2.64).100=55,56%
%Cu=44,44%%Cu=44,44%
a)\(n_{Al}=\dfrac{5,4}{27}=0,2mol\)
\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
0,2 0,3 0,1 0,3
b)\(V_{H_2}=0,3\cdot22,4=6,72l\)
c)\(n_{CuO}=\dfrac{32}{80}=0,4mol\)
\(CuO+H_2\rightarrow Cu+H_2O\)
0,4 0,3 0,3
\(m_{Cu}=0,3\cdot64=19,2g\)

nAl = 5.4/27 = 0.2 (mol)
2Al + 6HCl => 2AlCl3 + 3H2
0.2.......0.6......................0.3
CM HCl = 0.6 / 0.4 = 1.5 (M)
nCuO = 32/80 = 0.4 (mol)
CuO + H2 -to-> Cu + H2O
0.2.......0.2..........0.2
Chất rắn : 0.2 (mol) CuO dư , 0.2 (mol) Cu
%CuO =\(\dfrac{0,2.80}{0,2.80+0,2.64}\) 100% = 55.56%
%Cu = 44.44%
a) \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
b) \(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\)
Theo phương trình : \(n_{H_2}=\dfrac{3}{2}n_{Al}=0,3\left(mol\right)\)
\(\rightarrow V_{H_2}\left(đktc\right)=0,3.22,4=6,72\left(l\right)\)
c) Chất rắn : \(0,2\left(mol\right)\)
CuO dư : \(0,2\left(mol\right)Cu\)
\(\%CuO=\dfrac{0,2.80}{\left(0,2.80+0,2.64\right)}.100=55,56\%\)
\(\%Cu=44,44\%\)

\(n_{Fe}=\dfrac{16.8}{56}=0.3\left(mol\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(0.3....................0.3.........0.3\)
\(m_{FeCl_2}=0.3\cdot127=38.1\left(g\right)\)
\(V_{H_2}=0.6\cdot22.4=6.72\left(l\right)\)
\(CuO+H_2\underrightarrow{t^0}Cu+H_2O\)
\(.......0.3...0.3\)
\(m_{Cu}=0.3\cdot64=19.2\left(g\right)\)
PT: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
a, Ta có: \(n_{Fe}=\dfrac{16,8}{56}=0,3\left(mol\right)\)
Theo PT: \(n_{FeCl_2}=n_{Fe}=0,3\left(mol\right)\)
\(\Rightarrow m_{FeCl_2}=0,3.127=38,1\left(g\right)\)
b, Theo PT: \(n_{H_2}=n_{Fe}=0,3\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,3.22,4=6,72\left(l\right)\)
c, PT: \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
Theo PT: \(n_{Cu}=n_{H_2}=0,3\left(mol\right)\)
\(\Rightarrow m_{Cu}=0,3.64=19,2\left(g\right)\)
Bạn tham khảo nhé!

a. \(n_{Zn}=\dfrac{6.5}{65}=0,1\left(mol\right)\)
PTHH : Zn + 2HCl -> ZnCl2 + H2
0,1 0,2 0,1
b. \(V_{H_2}=0,1.22,4=2,24\left(l\right)\)
c. \(m_{HCl}=0,2.36,5=7,3\left(g\right)\)
\(n_{Zn}=\dfrac{6,5}{65}=0,1mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,1 0,2 0,1
\(V_{H_2}=0,1\cdot22,4=2,24l\)
\(m_{HCl}=0,2\cdot36,5=7,3g\)

a)
\(n_{Fe}=\dfrac{22,4}{56}=0,4\left(mol\right)\)
\(n_{HCl}=\dfrac{18,25}{36,5}=0,5\left(mol\right)\)
PTHH: Fe + 2HCl --> FeCl2 + H2
Xét tỉ lệ: \(\dfrac{0,4}{1}>\dfrac{0,5}{2}\) => Fe dư, HCl hết
PTHH: Fe + 2HCl --> FeCl2 + H2
0,5----------->0,25
=> VH2 = 0,25.22,4 = 5,6 (l)
b)\(n_{Fe_3O_4}=\dfrac{13,92}{232}=0,06\left(mol\right)\)
PTHH: Fe3O4 + 4H2 --to--> 3Fe + 4H2O
Xét tỉ lệ: \(\dfrac{0,06}{1}< \dfrac{0,25}{4}\) => Fe3O4 hết, H2 dư
PTHH: Fe3O4 + 4H2 --to--> 3Fe + 4H2O
0,06-->0,24------->0,18-->0,24
=> \(\left\{{}\begin{matrix}m_{Fe}=0,18.56=10,08\left(g\right)\\m_{H_2O}=0,24.18=4,32\left(g\right)\\m_{H_2\left(dư\right)}=\left(0,25-0,24\right).2=0,02\left(g\right)\end{matrix}\right.\)
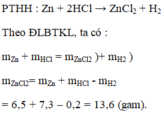
\(1,\\ Mg+2HCl\rightarrow MgCl_2+H_2\\ 2,\\ n_{HCl}=\dfrac{18,25}{36,5}=0,5\left(mol\right)\Rightarrow n_{Mg}=n_{MgCl_2}=n_{H_2}=\dfrac{0,5}{2}=0,25\left(mol\right)\\ V_{H_2\left(đktc\right)}=0,25.22,4=5,6\left(l\right)\\ 3,\\ m_{MgCl_2}=95.0,25=23,75\left(g\right)\\ 4,\\ H_2+CuO\rightarrow\left(t^o\right)Cu+H_2O\\ n_{Cu}=n_{H_2}=0,25\left(g\right)\\ m_{Cu}=0,25.64=16\left(g\right)\)
1. \(Mg+2HCl\rightarrow MgCl_2+H_2\)
2. \(n_{HCl}=\dfrac{m_{HCl}}{M_{HCl}}=\dfrac{18,25}{36,5}\approx0,5\left(mol\right)\)
Theo PTHH: \(n_{H_2}=\dfrac{1}{2}n_{HCl}\)
\(\Rightarrow n_{H_2}=\dfrac{1}{2}.0,5=0,25\left(mol\right)\)
\(\Rightarrow V_{H_2}=n_{H_2}.22,4=0,25.22,4=5,6\left(l\right)\)
3. Theo PTHH: \(n_{MgCl_2}=\dfrac{1}{2}n_{HCl}\)
\(\Rightarrow n_{MgCl_2}=0,25\left(mol\right)\)
\(m_{MgCl_2}=n_{MgCl_2}.M_{MgCl_2}=0,25.95=23,75\left(g\right)\)
4. \(H_2+CuO\rightarrow Cu+H_2O\)
Theo PTHH: \(n_{Cu}=n_{H_2}=0,25\left(mol\right)\)
\(\Rightarrow m_{Cu}=n_{Cu}.M_{Cu}=0,25.64=16\left(g\right)\)