Cho 200ml rượu Etylic 11,5 độ tác dụng bao nhiêu gam dung dịch CH3COOH 15%.Lượng este thu được đủ tác dụng bao nhiêu gam dung dịch NaOH 20%
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


a, \(n_{CH_3COOH}=0,2.1=0,2\left(mol\right)\)
PT: \(Mg+2CH_3COOH\rightarrow\left(CH_3COO\right)_2Mg+H_2\)
Theo PT: \(n_{Mg}=n_{H_2}=\dfrac{1}{2}n_{CH_3COOH}=0,1\left(mol\right)\)
\(\Rightarrow m=m_{Mg}=0,1.24=2,4\left(g\right)\)
\(V=V_{H_2}=0,1.22,4=2,24\left(l\right)\)
b, \(C_2H_5OH+O_2\underrightarrow{^{mengiam}}CH_3COOH+H_2O\)
Theo PT: \(n_{C_2H_5OH}=n_{CH_3COOH}=0,2\left(mol\right)\)
\(\Rightarrow m_{C_2H_5OH}=0,2.46=9,2\left(g\right)\)
\(\Rightarrow V_{ddC_2H_5OH}=\dfrac{9,2}{0,8}=11,5\left(ml\right)\)

a) PTHH: \(Mg+2CH_3COOH\rightarrow\left(CH_3COO\right)_2Mg+H_2\uparrow\)
Ta có: \(n_{CH_3COOH}=0,2\cdot1=0,2\left(mol\right)\)
\(\Rightarrow n_{Mg}=n_{H_2}=0,1\left(mol\right)\) \(\Rightarrow\left\{{}\begin{matrix}m_{Mg}=0,1\cdot24=2,4\left(g\right)\\V_{H_2}=0,1\cdot22,4=2,24\left(l\right)\end{matrix}\right.\)
b) PTHH: \(C_2H_5OH+O_2\xrightarrow[]{men}CH_3COOH+H_2O\)
Theo PTHH: \(n_{C_2H_5OH}=n_{CH_3COOH}=0,2\left(mol\right)\)
\(\Rightarrow m_{ddC_2H_5OH}=\dfrac{0,2\cdot46}{8\%}=115\left(g\right)\) \(\Rightarrow V_{C_2H_5OH}=\dfrac{115}{0,8}=143,75\left(ml\right)\)

a, nNaOH = 0,2.1 = 0,2 (mol)
PTHH: CH3COOH + NaOH ---> CH3COONa + H2O
0,2<---------0,2
=> \(\left\{{}\begin{matrix}m_{CH_3COOH}=0,2.60=12\left(g\right)\\m_{C_2H_5OH}=25,8-12=13,8\left(g\right)\end{matrix}\right.\)
=> \(\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{12}{25,8}.100\%=46,5\%\\\%m_{C_2H_5OH}=100\%-46,5\%=53,5\%\end{matrix}\right.\)
b, \(\left\{{}\begin{matrix}n_{CH_3COOH}=\dfrac{6,45}{25,8}.0,2=0,05\left(mol\right)\\n_{C_2H_5OH}=\dfrac{6,45-0,05.60}{46}=0,075\left(mol\right)\end{matrix}\right.\)
PTHH: \(CH_3COOH+C_2H_5OH\xrightarrow[t^o]{H_2SO_{4\left(đ\right)}}CH_3COOC_2H_5+H_2O\)
LTL: 0,05 < 0,075 => Rượu dư
=> \(n_{CH_3COOC_2H_5\left(LT\right)}=n_{CH_3COOH}=0,05\left(mol\right)\\ \)
=> \(m_{CH_3COOC_2H_5\left(TT\right)}=0,05.88.80\%=3,52\left(g\right)\)
a.\(n_{NaOH}=0,2.1=0,2mol\)
\(CH_3COOH+NaOH\rightarrow CH_3COONa+H_2O\)
0,2 0,2 ( mol )
\(\rightarrow\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,2.60}{25,8}=46,51\%\\\%m_{C_2H_5OH}=100\%-46,51\%=53,49\%\end{matrix}\right.\)
b.Bạn check lại đề giúp mình:((

\(n_{CH_3COOH}=\dfrac{300\cdot5\%}{60}=0.25\left(mol\right)\)
\(2CH_3COOH+Zn\rightarrow\left(CH_3COO\right)_2Zn+H_2\)
\(0.25........................................................0.125\)
\(V_{H_2}=0.125\cdot22.4=2.8\left(l\right)\)
\(n_{C_2H_5OH}=0.1\cdot2=0.2\left(mol\right)\)
\(CH_3COOH+C_2H_5OH⇌CH_3COOC_2H_5+H_2O\left(ĐK:H_2SO_{4\left(đ\right)},t^0\right)\)
\(0.2......................0.2.....................0.2\)
\(\Rightarrow CH_3COOHdư\)
\(m_{CH_3COOC_2H_5}=0.2\cdot88=17.6\left(g\right)\)

a) n CH3COOH = 300.5%/60 = 0,25(mol)
Zn + 2CH3COOH $\to$ (CH3COO)2Zn + H2
Theo PTHH :
n H2 = 1/2 n CH3COOH = 0,25/2 = 0,125(mol)
V H2 = 0,125.22,4 = 2,8(lít)
b) n C2H5OH = 0,1.2 = 0,2(mol)
\(CH_3COOH + C_2H_5OH \buildrel{{H_2SO_4,t^o}}\over\rightleftharpoons CH_3COOC_2H_5 + H_2O\)
Ta thấy :
n CH3COOH = 0,25 > n C2H5OH = 0,2 => CH3COOH dư
n CH3COOC2H5 = n C2H5OH = 0,2 mol
=> m CH3COOC2H5 = 0,2.88 = 17,6 gam

a, \(n_{CH_3COOH}=\dfrac{15}{60}=0,25\left(mol\right)\)
PT: \(CH_3COOH+NaOH\rightarrow CH_3COONa+H_2O\)
Theo PT: \(n_{CH_3COONa}=n_{CH_3COOH}=0,25\left(mol\right)\)
\(\Rightarrow a=m_{CH_3COONa}=0,25.82=20,5\left(g\right)\)
b, \(m_{dd}=\dfrac{15}{2\%}=750\left(g\right)\)
\(n_{CH_3COOH}=\dfrac{15}{60}=0,25\left(mol\right)\\ NaOH+CH_3COOH\rightarrow CH_3COONa+H_2O\\a, n_{CH_3COONa}=n_{CH_3COOH}=0,25\left(mol\right)\\ a=m_{CH_3COONa}=0,25.82=20,5\left(g\right)\\ b,m_{ddCH_3COOH}=\dfrac{15.100}{2}=750\left(g\right)\)

\(a,n_{CH_3COOH}=\dfrac{120.20}{100}=24\left(g\right)\\ \rightarrow n_{CH_3COOH}=\dfrac{24}{60}=0,4\left(mol\right)\)
PTHH: \(2CH_3COOH+Na_2CO_3\rightarrow2CH_3COONa+CO_2\uparrow+H_2O\)
0,4----------->0,2-------------->0,4-------------->0,2
\(\rightarrow m_{ddNa_2CO_3}=\dfrac{0,2.106}{10\%}=212\left(g\right)\)
\(\rightarrow m_{ddA}=212+120-0,2.44=323,2\left(g\right)\\ \rightarrow C\%_{CH_3COONa}=\dfrac{0,4.82}{323,2}.100\%=10,15\%\)
b, PTHH: \(C_2H_5OH+O_2\underrightarrow{\text{men giấm}}CH_3COOH+H_2O\)
0,4<------------------------0,4
\(\rightarrow V_{ddC_2H_5OH}=\dfrac{0,4.46.100}{0,8.46}=50\left(ml\right)\)

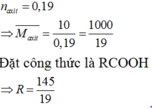

\(D=0,8g\)/cm3
Trong 200ml rượu etylic \(11,5^o\) có:
\(V_{C_2H_5OH}=V_{dd}\cdot\dfrac{11,5}{100}=200\cdot\dfrac{11,5}{100}=23ml\)
\(\Rightarrow m_{C_2H_5OH}=D\cdot V=23\cdot0,8=18,4g\)
\(C_2H_5OH+O_2\rightarrow CH_3COOH+H_2O\)
46 60 (gam)
18,4 m (gam)
\(\Rightarrow m=24g\)
\(m_{ddCH_3COOH}=\dfrac{24}{15\%}\cdot100\%=160g\)